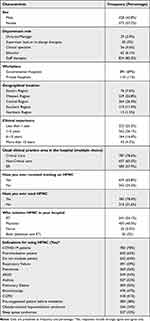
James SLAD, Abate D, Abate KH, Abay SM, Abbafati C, Abbasi N, et al. Global, regional, and national incidence, prevalence, and years lived with disability for 354 diseases and injuries for 195 countries and territories, 1990–2017: a systematic analysis for the global burden of disease study 2017. Lancet. 2018;392(10159):1789–858.
Article
Google Scholar
Mattila T, Vasankari T, Kauppi P, Mazur W, Härkänen T, Heliövaara M. Mortality of asthma, COPD, and asthma-COPD overlap during an 18 year follow up. Respir Med. 2023;207: 107112.
Article
PubMed
Google Scholar
Hogg JC. Pathophysiology of airflow limitation in chronic obstructive pulmonary disease. Lancet. 2004;364(9435):709–21.
Article
PubMed
Google Scholar
Postma DS, Rabe KF. The asthma-COPD overlap syndrome. N Engl J Med. 2015;373(13):1241–9.
Article
CAS
PubMed
Google Scholar
Marcon A, Locatelli F, Dharmage SC, Svanes C, Heinrich J, Leynaert B, et al. The coexistence of asthma and COPD: risk factors, clinical history and lung function trajectories. Eur Respir J. 2021. doi.org/10.1183/13993003.04656-2020.
Article
PubMed
PubMed Central
Google Scholar
Slats A, Taube C. Asthma and chronic obstructive pulmonary disease overlap: asthmatic chronic obstructive pulmonary disease or chronic obstructive asthma? Ther Adv Respir Dis. 2016;10(1):57–71.
Article
PubMed
Google Scholar
Çolak Y. Undiagnosed (or unrecognised) COPD and asthma: does active case-finding identify clinically impaired patients with treatment potential. Am J Respir Crit Care Med. 2023. doi.org/10.1164/rccm.202310-1793ED.
Article
PubMed
PubMed Central
Google Scholar
Çolak Y, Afzal S, Nordestgaard BG, Vestbo J, Lange P. Prognosis of asymptomatic and symptomatic, undiagnosed COPD in the general population in Denmark: a prospective cohort study. Lancet Respir Med. 2017;5(5):426–34.
Article
PubMed
Google Scholar
Dasgupta S, Ghosh N, Bhattacharyya P, Roy Chowdhury S, Chaudhury K. Metabolomics of asthma, COPD, and asthma-COPD overlap: an overview. Crit Rev Clin Lab Sci. 2023;60(2):153–70.
Article
PubMed
Google Scholar
Kelly RS, Dahlin A, McGeachie MJ, Qiu W, Sordillo J, Wan ES, et al. Asthma metabolomics and the potential for integrative omics in research and the clinic. Chest. 2017;151(2):262–77.
Article
PubMed
Google Scholar
Terracciano R, Preianò M, Palladino GP, Carpagnano GE, Barbaro MP, Pelaia G, et al. Peptidome profiling of induced sputum by mesoporous silica beads and MALDI-TOF MS for non-invasive biomarker discovery of chronic inflammatory lung diseases. Proteomics. 2011;11(16):3402–14.
Article
CAS
PubMed
Google Scholar
Pelaia G, Terracciano R, Vatrella A, Gallelli L, Busceti MT, Calabrese C, Stellato C, Savino R, Maselli R. Application of proteomics and peptidomics to COPD. Biomed Res Int. 2014;2014: 764581.
Article
PubMed
PubMed Central
Google Scholar
Gao D, Zhang L, Song D, Lv J, Wang L, Zhou S, et al. Values of integration between lipidomics and clinical phenomes in patients with acute lung infection, pulmonary embolism, or acute exacerbation of chronic pulmonary diseases: a preliminary study. J Transl Med. 2019;17(1):162.
Article
PubMed
PubMed Central
Google Scholar
Qiu S, Cai Y, Yao H, Lin C, Xie Y, Tang S, et al. Small molecule metabolites: discovery of biomarkers and therapeutic targets. Signal Transduct Target Ther. 2023;8(1):132.
Article
PubMed
PubMed Central
Google Scholar
Chen L, Lu W, Wang L, Xing X, Chen Z, Teng X, et al. Metabolite discovery through global annotation of untargeted metabolomics data. Nat Method. 2021;18(11):1377–85.
Article
Google Scholar
Luan H, Gu W, Li H, Wang Z, Lu L, Ke M, et al. Serum metabolomic and lipidomic profiling identifies diagnostic biomarkers for seropositive and seronegative rheumatoid arthritis patients. J Transl Med. 2021;19(1):500.
Article
CAS
PubMed
PubMed Central
Google Scholar
Wang R, Li B, Lam SM, Shui G. Integration of lipidomics and metabolomics for in-depth understanding of cellular mechanism and disease progression. J Genet Genom. 2020;47(2):69–83.
Article
CAS
Google Scholar
Correnti S, Preianò M, Fregola A, Gamboni F, Stephenson D, Savino R, et al. Seminal plasma untargeted metabolomic and lipidomic profiling for the identification of a novel panel of biomarkers and therapeutic targets related to male infertility. Front Pharmacol. 2023;14:1275832.
Article
CAS
PubMed
PubMed Central
Google Scholar
Liang Y, Gai XY, Chang C, Zhang X, Wang J, Li TT. Metabolomic profiling differences among asthma, COPD, and healthy subjects: A LC-MS-based metabolomic analysis. Biomed Environ Sci. 2019;32(9):659–72.
CAS
PubMed
Google Scholar
Adamko DJ, Nair P, Mayers I, Tsuyuki RT, Regush S, Rowe BH. Metabolomic profiling of asthma and chronic obstructive pulmonary disease: a pilot study differentiating diseases. J Allerg Clin Immunol. 2015;136(3):571-80.e3.
Article
Google Scholar
Khamis MM, Holt T, Awad H, El-Aneed A, Adamko DJ. Comparative analysis of creatinine and osmolality as urine normalization strategies in targeted metabolomics for the differential diagnosis of asthma and COPD. Metabolomics. 2018;14(9):115.
Article
PubMed
Google Scholar
D’Amato M, Iadarola P, Viglio S. Proteomic analysis of human sputum for the diagnosis of lung disorders: where are we today? Int J Mol Sci. 2022. doi.org/10.3390/ijms23105692.
Article
PubMed
PubMed Central
Google Scholar
Global Strategy for Asthma Management and Prevention. ginasthma.org/wp-content/uploads/2019/06/GINA-2019-main-report-June-2019-wms.pdf. Accessed on 14 December 2023.
Global Strategy for Prevention, Diagnosis and Management of COPD. goldcopd.org/wp-content/uploads/2023/03/GOLD-2023-ver-1.3-17Feb2023_WMV.pdf. Accessed on 14 December 2023.
Nemkov T, Reisz JA, Gehrke S, Hansen KC, D’Alessandro A. High-throughput metabolomics: isocratic and gradient mass spectrometry-based methods. Method Mol Biol. 2019;1978:13–26.
Article
CAS
Google Scholar
Reisz JA, Zheng C, D’Alessandro A, Nemkov T. Untargeted and semi-targeted lipid analysis of biological samples using mass spectrometry-based metabolomics. Method Mol Biol. 2019;1978:121–35.
Article
CAS
Google Scholar
Barnes PJ. Cellular and molecular mechanisms of asthma and COPD. Clin Sci. 2017;131(13):1541–58.
Article
CAS
Google Scholar
Han MK, Agusti A, Calverley PM, Celli BR, Criner G, Curtis JL, et al. Chronic obstructive pulmonary disease phenotypes: the future of COPD. Am J Respir Crit Care Med. 2010;182(5):598–604.
Article
PubMed
PubMed Central
Google Scholar
Wenzel SE. Complex phenotypes in asthma: current definitions. Pulm Pharmacol Ther. 2013;26(6):710–5.
Article
CAS
PubMed
Google Scholar
Nickler M, Ottiger M, Steuer C, Huber A, Anderson JB, Müller B, et al. Systematic review regarding metabolic profiling for improved pathophysiological understanding of disease and outcome prediction in respiratory infections. Respir Res. 2015;16:125.
Article
PubMed
PubMed Central
Google Scholar
Sim S, Choi Y, Park HS. Potential metabolic biomarkers in adult asthmatics. Metabolites. 2021. doi.org/10.3390/metabo11070430.
Article
PubMed
PubMed Central
Google Scholar
Pulik K, Mycroft K, Korczyński P, Ciechanowicz AK, Górska K. Metabolomic analysis of respiratory epithelial lining fluid in patients with chronic obstructive pulmonary disease—a systematic review. Cells. 2023. doi.org/10.3390/cells12060833.
Article
PubMed
PubMed Central
Google Scholar
Sinha A, Desiraju K, Aggarwal K, Kutum R, Roy S, Lodha R, et al. Exhaled breath condensate metabolome clusters for endotype discovery in asthma. J Transl Med. 2017;15(1):262.
Article
CAS
PubMed
PubMed Central
Google Scholar
Ravi A, Goorsenberg AWM, Dijkhuis A, Dierdorp BS, Dekker T, van Weeghel M, et al. Metabolic differences between bronchial epithelium from healthy individuals and patients with asthma and the effect of bronchial thermoplasty. J Allerg Clin Immunol. 2021;148(5):1236–48.
Article
CAS
Google Scholar
Maniscalco M, Paris D, Melck DJ, Molino A, Carone M, Ruggeri P, et al. Differential diagnosis between newly diagnosed asthma and COPD using exhaled breath condensate metabolomics: a pilot study. Eur Respir J. 2018;51(3):1701825.
Article
PubMed
Google Scholar
Fens N, Roldaan AC, van der Schee MP, Boksem RJ, Zwinderman AH, Bel EH, et al. External validation of exhaled breath profiling using an electronic nose in the discrimination of asthma with fixed airways obstruction and chronic obstructive pulmonary disease. Clin Exp Allerg. 2011;41(10):1371–8.
Article
CAS
Google Scholar
Jiang T, Dai L, Li P, Zhao J, Wang X, An L, et al. Lipid metabolism and identification of biomarkers in asthma by lipidomic analysis. Biochim Biophys Acta Mol Cell Biol Lipid. 2021;1866(2): 158853.
Article
CAS
Google Scholar
Ntontsi P, Ntzoumanika V, Loukides S, Benaki D, Gkikas E, Mikros E, et al. EBC metabolomics for asthma severity. J Breath Res. 2020;14(3): 036007.
Article
CAS
PubMed
Google Scholar
Ried JS, Baurecht H, Stückler F, Krumsiek J, Gieger C, Heinrich J, et al. Integrative genetic and metabolite profiling analysis suggests altered phosphatidylcholine metabolism in asthma. Allergy. 2013;68(5):629–36.
Article
CAS
PubMed
Google Scholar
Tian M, Chen M, Bao YL, Xu CD, Qin QZ, Zhang WX, et al. Sputum metabolomic profiling of bronchial asthma based on quadruple time-of-flight mass spectrometry. Int J Clin Exp Pathol. 2017;10(10):10363–73.
PubMed
PubMed Central
Google Scholar
Quinn KD, Schedel M, Nkrumah-Elie Y, Joetham A, Armstrong M, Cruickshank-Quinn C, et al. Dysregulation of metabolic pathways in a mouse model of allergic asthma. Allergy. 2017;72(9):1327–37.
Article
CAS
PubMed
Google Scholar
Ran N, Pang Z, Gu Y, Pan H, Zuo X, Guan X, et al. An updated overview of metabolomic profile changes in chronic obstructive pulmonary disease. Metabolites. 2019;9(6):111.
Article
CAS
PubMed
PubMed Central
Google Scholar
Pacheco-Alvarez D, Solórzano-Vargas RS, Del Río AL. Biotin in metabolism and its relationship to human disease. Arch Med Res. 2002;33(5):439–47.
Article
CAS
PubMed
Google Scholar
Vaz FM, Wanders RJ. Carnitine biosynthesis in mammals. Biochem J. 2002;361(Pt 3):417–29.
Article
CAS
PubMed
PubMed Central
Google Scholar
Halper-Stromberg E, Gillenwater L, Cruickshank-Quinn C, O’Neal WK, Reisdorph N, Petrache I, et al. Bronchoalveolar lavage Fluid from COPD patients reveals more compounds associated with disease than matched plasma. Metabolites. 2019;9(8):157.
Article
CAS
PubMed
PubMed Central
Google Scholar
Cruickshank-Quinn CI, Jacobson S, Hughes G, Powell RL, Petrache I, Kechris K, et al. Metabolomics and transcriptomics pathway approach reveals outcome-specific perturbations in COPD. Sci Rep. 2018;8(1):17132.
Article
PubMed
PubMed Central
Google Scholar
Ghosh N, Choudhury P, Subramani E, Saha D, Sengupta S, Joshi M, et al. Metabolomic signatures of asthma-COPD overlap (ACO) are different from asthma and COPD. Metabolomics. 2019;15(6):87.
Article
CAS
PubMed
Google Scholar
Sagar NA, Tarafdar S, Agarwal S, Tarafdar A, Sharma S. Polyamines: functions, metabolism, and role in human disease management. Med Sci. 2021;9(2):44.
CAS
Google Scholar
Jain V. Role of polyamines in asthma pathophysiology. Med Sci. 2018;6(1):4.
Google Scholar
Hoet PH, Nemery B. Polyamines in the lung: polyamine uptake and polyamine-linked pathological or toxicological conditions. Am J Physiol Lung Cell Mol Physiol. 2000;278(3):L417-33.
Article
CAS
PubMed
Google Scholar
Kurosawa M, Shimizu Y, Tsukagoshi H, Ueki M. Elevated levels of peripheral-blood, naturally occurring aliphatic polyamines in bronchial asthmatic patients with active symptoms. Allergy. 1992;47(6):638–43.
Article
CAS
PubMed
Google Scholar
Zimmermann N, King NE, Laporte J, Yang M, Mishra A, Pope SM, et al. Dissection of experimental asthma with DNA microarray analysis identifies arginase in asthma pathogenesis. J Clin Invest. 2003;111(12):1863–74.
Article
CAS
PubMed
PubMed Central
Google Scholar
Kurosawa M, Uno D, Kobayashi S. Naturally occurring aliphatic polyamines-induced histamine release from rat peritoneal mast cells. Allerg. 1991;46(5):349–54.
Article
CAS
Google Scholar
Ilmarinen P, Moilanen E, Erjefält JS, Kankaanranta H. The polyamine spermine promotes survival and activation of human eosinophils. J Allerg Clin Immunol. 2015;136(2):482-4.e11.
Article
CAS
Google Scholar
North ML, Grasemann H, Khanna N, Inman MD, Gauvreau GM, Scott JA. Increased ornithine-derived polyamines cause airway hyperresponsiveness in a mouse model of asthma. Am J Respir Cell Mol Biol. 2013;48(6):694–702.
Article
CAS
PubMed
Google Scholar
Zuo L, Koozechian MS, Chen LL. Characterization of reactive nitrogen species in allergic asthma. Ann Allerg Asthma Immunol. 2014;112(1):18–22.
Article
CAS
Google Scholar
Ghosh S, Erzurum SC. Nitric oxide metabolism in asthma pathophysiology. Biochim Biophys Acta. 2011;1810(11):1008–16.
Article
CAS
PubMed
PubMed Central
Google Scholar
Maarsingh H, Leusink J, Zaagsma J, Meurs H. Role of the l-citrulline/l-arginine cycle in iNANC nerve-mediated nitric oxide production and airway smooth muscle relaxation in allergic asthma. Eur J Pharmacol. 2006;546(1–3):171–6.
Article
CAS
PubMed
Google Scholar
Wong JM, de Souza R, Kendall CW, Emam A, Jenkins DJ. Colonic health: fermentation and short chain fatty acids. J Clin Gastroenterol. 2006;40(3):235–43.
Article
CAS
PubMed
Google Scholar
Rajendiran E, Ramadass B, Ramprasath V. Understanding connections and roles of gut microbiome in cardiovascular diseases. Can J Microbiol. 2021;67(2):101–11.
Article
CAS
PubMed
Google Scholar
Enaud R, Prevel R, Ciarlo E, Beaufils F, Wieërs G, Guery B, et al. The gut-lung axis in health and respiratory diseases: a place for inter-organ and inter-kingdom crosstalks. Front Cell Infect Microbiol. 2020;10:9.
Article
CAS
PubMed
PubMed Central
Google Scholar
Anand S, Mande SS. Diet, Microbiota and Gut-Lung Connection. Front Microbiol. 2018;9:2147.
Article
PubMed
PubMed Central
Google Scholar
Ghorbani P, Santhakumar P, Hu Q, Djiadeu P, Wolever TM, Palaniyar N, et al. Short-chain fatty acids affect cystic fibrosis airway inflammation and bacterial growth. Eur Respir J. 2015;46(4):1033–45.
Article
CAS
PubMed
Google Scholar
Liu Q, Tian X, Maruyama D, Arjomandi M, Prakash A. Lung immune tone via gut-lung axis: gut-derived LPS and short-chain fatty acids’ immunometabolic regulation of lung IL-1β, FFAR2, and FFAR3 expression. Am J Physiol Lung Cell Mol Physiol. 2021;321(1):L65-l78.
Article
CAS
PubMed
PubMed Central
Google Scholar
Yoon HJ, Park MK, Lee H, Park TS, Park DW, Moon JY, et al. Effects of respiratory short-chain fatty acids on bronchial inflammation in asthma. World Allerg Organ J. 2020;13(8):100204.
Article
Google Scholar
Richards LB, Li M, Folkerts G, Henricks PAJ, Garssen J, van Esch B. Butyrate and propionate restore the cytokine and house dust mite compromised barrier function of human bronchial airway epithelial cells. Int J Mol Sci. 2020;22(1):65.
Article
PubMed
PubMed Central
Google Scholar
Tatsuta M, Kan OK, Ishii Y, Yamamoto N, Ogawa T, Fukuyama S, et al. Effects of cigarette smoke on barrier function and tight junction proteins in the bronchial epithelium: protective role of cathelicidin LL-37. Respir Res. 2019;20(1):251.
Article
PubMed
PubMed Central
Google Scholar
Li N, Dai Z, Wang Z, Deng Z, Zhang J, Pu J, et al. Gut microbiota dysbiosis contributes to the development of chronic obstructive pulmonary disease. Respir Res. 2021;22(1):274.
Article
CAS
PubMed
PubMed Central
Google Scholar
Li C, Zhao H. Tryptophan and its metabolites in lung cancer: basic functions and clinical significance. Front Oncol. 2021;11: 707277.
Article
CAS
PubMed
PubMed Central
Google Scholar
Naz S, Bhat M, Ståhl S, Forsslund H, Sköld CM, Wheelock ÅM, et al. Dysregulation of the tryptophan pathway evidences gender differences in COPD. Metabolites. 2019;9(10):212.
Article
CAS
PubMed
PubMed Central
Google Scholar
Gulcev M, Reilly C, Griffin TJ, Broeckling CD, Sandri BJ, Witthuhn BA, et al. Tryptophan catabolism in acute exacerbations of chronic obstructive pulmonary disease. Int J Chron Obstruct Pulmon Dis. 2016;11:2435–46.
Article
CAS
PubMed
PubMed Central
Google Scholar
Mellor AL, Munn DH. IDO expression by dendritic cells: tolerance and tryptophan catabolism. Nat Rev Immunol. 2004;4(10):762–74.
Article
CAS
PubMed
Google Scholar
Yan Z, Chen B, Yang Y, Yi X, Wei M, Ecklu-Mensah G, et al. Multi-omics analyses of airway host-microbe interactions in chronic obstructive pulmonary disease identify potential therapeutic interventions. Nat Microbiol. 2022;7(9):1361–75.
Article
CAS
PubMed
Google Scholar
Ubhi BK, Cheng KK, Dong J, Janowitz T, Jodrell D, Tal-Singer R, et al. Targeted metabolomics identifies perturbations in amino acid metabolism that sub-classify patients with COPD. Mol Biosyst. 2012;8(12):3125–33.
Article
CAS
PubMed
Google Scholar
Maneechotesuwan K, Kasetsinsombat K, Wongkajornsilp A, Barnes PJ. Decreased indoleamine 2,3-dioxygenase activity and IL-10/IL-17A ratio in patients with COPD. Thorax. 2013;68(4):330–7.
Article
PubMed
Google Scholar
Liu H, Liu L, Fletcher BS, Visner GA. Novel action of indoleamine 2,3-dioxygenase attenuating acute lung allograft injury. Am J Respir Crit Care Med. 2006;173(5):566–72.
Article
CAS
PubMed
Google Scholar
Reyes Ocampo J, Lugo Huitrón R, González-Esquivel D, Ugalde-Muñiz P, Jiménez-Anguiano A, Pineda B, et al. Kynurenines with neuroactive and redox properties: relevance to aging and brain diseases. Oxid Med Cell Longev. 2014;2014: 646909.
Article
PubMed
PubMed Central
Google Scholar
Savonije K, Weaver DF. The role of tryptophan metabolism in alzheimer’s disease. Brain Sci. 2023;13(2):292.
Article
CAS
PubMed
PubMed Central
Google Scholar
Sorgdrager FJH, Naudé PJW, Kema IP, Nollen EA, Deyn PP. Tryptophan metabolism in inflammaging from biomarker to therapeutic target. Front Immunol. 2019;10:2565.
Article
CAS
PubMed
PubMed Central
Google Scholar
Kalantar-Zadeh K, Ganz T, Trumbo H, Seid MH, Goodnough LT, Levine MA. Parenteral iron therapy and phosphorus homeostasis: a review. Am J Hematol. 2021;96(5):606–16.
Article
CAS
PubMed
PubMed Central
Google Scholar
Li S, Huang Q, Nan W, He B. Association between serum phosphate and in-hospital mortality of patients with AECOPD: a retrospective analysis on eICU database. Heliyon. 2023;9(9): e19748.
Article
PubMed
PubMed Central
Google Scholar
Jung SY, Kwon J, Park S, Jhee JH, Yun HR, Kim H, et al. Phosphate is a potential biomarker of disease severity and predicts adverse outcomes in acute kidney injury patients undergoing continuous renal replacement therapy. PLoS ONE. 2018;13(2): e0191290.
Article
PubMed
PubMed Central
Google Scholar
Xu H, Evans M, Gasparini A, Szummer K, Spaak J, Ärnlöv J, et al. Outcomes associated to serum phosphate levels in patients with suspected acute coronary syndrome. Int J Cardiol. 2017;245:20–6.
Article
PubMed
Google Scholar
Stroda A, Brandenburg V, Daher A, Cornelissen C, Goettsch C, Keszei A, et al. Serum phosphate and phosphate-regulatory hormones in COPD patients. Respir Res. 2018;19(1):183.
Article
PubMed
PubMed Central
Google Scholar
Farah R, Khamisy-Farah R, Arraf Z, Jacobson L, Makhoul N. Hypophosphatemia as a prognostic value in acute exacerbation of COPD. Clin Respir J. 2013;7(4):407–15.
Article
CAS
PubMed
Google Scholar
Campos-Obando N, Lahousse L, Brusselle G, Stricker BH, Hofman A, Franco OH, et al. Serum phosphate levels are related to all-cause, cardiovascular and COPD mortality in men. Eur J Epidemiol. 2018;33(9):859–71.
Article
CAS
PubMed
PubMed Central
Google Scholar
Giraud MF, Naismith JH. The rhamnose pathway. Curr Opin Struct Biol. 2000;10(6):687–96.
Article
CAS
PubMed
Google Scholar
Melamed J, Kocev A, Torgov V, Veselovsky V, Brockhausen I. Biosynthesis of the Pseudomonas aeruginosa common polysaccharide antigen by d-Rhamnosyltransferases WbpX and WbpY. Glycoconj J. 2022;39(3):393–11.
Article
CAS
PubMed
PubMed Central
Google Scholar
Eklöf J, Sørensen R, Ingebrigtsen TS, Sivapalan P, Achir I, Boel JB, et al. Pseudomonas aeruginosa and risk of death and exacerbations in patients with chronic obstructive pulmonary disease: an observational cohort study of 22 053 patients. Clin Microbiol Infect. 2020;26(2):227–34.
Article
PubMed
Google Scholar
Jacobs DM, Ochs-Balcom HM, Noyes K, Zhao J, Leung WY, Pu CY, et al. Impact of pseudomonas aeruginosa isolation on mortality and outcomes in an outpatient chronic obstructive pulmonary disease cohort. Open Forum Infect Dis. 2020;7(1):ofz546.
Article
PubMed
PubMed Central
Google Scholar
Zhang Q, Illing R, Hui CK, Downey K, Carr D, Stearn M, et al. Bacteria in sputum of stable severe asthma and increased airway wall thickness. Respir Res. 2012;13(1):35.
Article
PubMed
PubMed Central
Google Scholar
Garcia-Clemente M, de la Rosa D, Máiz L, Girón R, Blanco M, Olveira C, et al. Impact of pseudomonas aeruginosa infection on patients with chronic inflammatory airway diseases. J Clin Med. 2020;9(12):3800.
Article
PubMed
PubMed Central
Google Scholar
Kayongo A, Robertson NM, Siddharthan T, Ntayi ML, Ndawula JC, Sande OJ, et al. Airway microbiome-immune crosstalk in chronic obstructive pulmonary disease. Front Immunol. 2022;13:1085551.
Article
CAS
PubMed
Google Scholar
Mander A, Langton-Hewer S, Bernhard W, Warner JO, Postle AD. Altered phospholipid composition and aggregate structure of lung surfactant is associated with impaired lung function in young children with respiratory infections. Am J Respir Cell Mol Biol. 2002;27(6):714–21.
Article
CAS
PubMed
Google Scholar
Telenga ED, Hoffmann RF, Ruben tK, Hoonhorst SJ, Willemse BW, van Oosterhout AJ, et al. Untargeted lipidomic analysis in chronic obstructive pulmonary disease uncovering sphingolipids. Am J Respir Crit Care Med. 2014;190(2):155–64.
Article
CAS
PubMed
Google Scholar
Chen H, Li Z, Dong L, Wu Y, Shen H, Chen Z. Lipid metabolism in chronic obstructive pulmonary disease. Int J Chron Obstruct Pulmon Dis. 2019;14:1009–18.
Article
CAS
PubMed
PubMed Central
Google Scholar
Gai X, Guo C, Zhang L, Zhang L, Abulikemu M, Wang J, et al. Serum glycerophospholipid profile in acute exacerbation of chronic obstructive pulmonary disease. Front Physiol. 2021;12: 646010.
Article
PubMed
PubMed Central
Google Scholar
Kilk K, Aug A, Ottas A, Soomets U, Altraja S, Altraja A. Phenotyping of chronic obstructive pulmonary disease based on the integration of metabolomes and clinical characteristics. Int J Mol Sci. 2018;19(3):666.
Article
PubMed
PubMed Central
Google Scholar
Kang YP, Lee WJ, Hong JY, Lee SB, Park JH, Kim D, et al. Novel approach for analysis of bronchoalveolar lavage fluid (BALF) using HPLC-QTOF-MS-based lipidomics: lipid levels in asthmatics and corticosteroid-treated asthmatic patients. J Proteome Res. 2014;13(9):3919–29.
Article
CAS
PubMed
Google Scholar
Liu X, Zhang H, Si Y, Du Y, Wu J, Li J. High-coverage lipidomics analysis reveals biomarkers for diagnosis of acute exacerbation of chronic obstructive pulmonary disease. J Chromatogr B Analyt Technol Biomed Life Sci. 2022;1201–1202: 123278.
Article
PubMed
Google Scholar
Agudelo CW, Kumley BK, Area-Gomez E, Xu Y, Dabo AJ, Geraghty P, et al. Decreased surfactant lipids correlate with lung function in chronic obstructive pulmonary disease (COPD). PLoS ONE. 2020;15(2): e0228279.
Article
CAS
PubMed
PubMed Central
Google Scholar
Hannun YA, Obeid LM. Principles of bioactive lipid signalling: lessons from sphingolipids. Nat Rev Mol Cell Biol. 2008;9(2):139–50.
Article
CAS
PubMed
Google Scholar
Kim J, Suresh B, Lim MN, Hong SH, Kim KS, Song HE, et al. Metabolomics reveals dysregulated sphingolipid and amino acid metabolism associated with chronic obstructive pulmonary disease. Int J Chron Obstruct Pulmon Dis. 2022;17:2343–53.
Article
CAS
PubMed
PubMed Central
Google Scholar
Yang Y, Uhlig S. The role of sphingolipids in respiratory disease. Ther Adv Respir Dis. 2011;5(5):325–44.
Article
CAS
PubMed
Google Scholar
Bowler RP, Jacobson S, Cruickshank C, Hughes GJ, Siska C, Ory DS, et al. Plasma sphingolipids associated with chronic obstructive pulmonary disease phenotypes. Am J Respir Crit Care Med. 2015;191(3):275–84.
Article
PubMed
PubMed Central
Google Scholar
McCann MR, George De la Rosa MV, Rosania GR, Stringer KA. l-Carnitine and acylcarnitines: mitochondrial biomarkers for precision medicine. Metabolites. 2021;11(1):51.
Article
CAS
PubMed
PubMed Central
Google Scholar
Gillenwater LA, Kechris KJ, Pratte KA, Reisdorph N, Petrache I, Labaki WW, et al. Metabolomic profiling reveals sex specific associations with chronic obstructive pulmonary disease and emphysema. Metabolites. 2021;11(3):161.
Article
CAS
PubMed
PubMed Central
Google Scholar
Callejón-Leblic B, Pereira-Vega A, Vázquez-Gandullo E, Sánchez-Ramos JL, Gómez-Ariza JL, García-Barrera T. Study of the metabolomic relationship between lung cancer and chronic obstructive pulmonary disease based on direct infusion mass spectrometry. Biochimie. 2019;157:111–22.
Article
PubMed
Google Scholar
Kim DJ, Oh JY, Rhee CK, Park SJ, Shim JJ, Cho JY. Metabolic fingerprinting uncovers the distinction between the phenotypes of tuberculosis associated COPD and smoking-induced COPD. Front Med. 2021;8: 619077.
Article
Google Scholar
Naz S, Kolmert J, Yang M, Reinke SN, Kamleh MA, Snowden S, et al. Metabolomics analysis identifies sex-associated metabotypes of oxidative stress and the autotaxin-lysoPA axis in COPD. Eur Respir J. 2017;49(6):1602322.
Article
PubMed
PubMed Central
Google Scholar
Yu B, Flexeder C, McGarrah RW 3rd, Wyss A, Morrison AC, North KE, et al. Metabolomics identifies novel blood biomarkers of pulmonary function and COPD in the general population. Metabolites. 2019;9(4):61.
Article
CAS
PubMed
PubMed Central
Google Scholar
Lommatzsch M, Cicko S, Müller T, Lucattelli M, Bratke K, Stoll P, et al. Extracellular adenosine triphosphate and chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 2010;181(9):928–34.
Article
CAS
PubMed
Google Scholar
Idzko M, Hammad H, van Nimwegen M, Kool M, Willart MA, Muskens F, et al. Extracellular ATP triggers and maintains asthmatic airway inflammation by activating dendritic cells. Nat Med. 2007;13(8):913–9.
Article
CAS
PubMed
Google Scholar
Hunninghake GM, Cho MH, Tesfaigzi Y, Soto-Quiros ME, Avila L, Lasky-Su J, et al. MMP12, lung function, and COPD in high-risk populations. N Engl J Med. 2009;361(27):2599–608.
Article
CAS
PubMed
PubMed Central
Google Scholar
Reinke SN, Naz S, Chaleckis R, Gallart-Ayala H, Kolmert J, Kermani NZ, et al. Urinary metabotype of severe asthma evidences decreased carnitine metabolism independent of oral corticosteroid treatment in the U-BIOPRED study. Eur Respir J. 2022;59(6):2101733.
Article
CAS
PubMed
PubMed Central
Google Scholar
Terracciano R, Pelaia G, Preianò M, Savino R. Asthma and COPD proteomics: current approaches and future directions. Proteom Clin Appl. 2015;9(1–2):203–20.
Article
CAS
Google Scholar