Introduction
The high-flow nasal cannula (HFNC) has become an essential non-invasive oxygen therapy device. Recently, it has been used widely in clinical settings as a non-invasive respiratory support method to improve oxygenation in adult patients.1 HFNC has several advantages over conventional low-flow devices (eg, nasal cannula or simple face masks): enhanced patient comfort, increased humidification, improved secretion clearance and reduced effort in breathing.2,3 The HFNC system is a unique device that is simple and easy to use, needing only an active heated humidifier, flow generator, single circuit, and soft nasal cannula.4 This device was developed to maintain high oxygen flow and improve the efficiency of ventilation by delivering warmed and humidified oxygen with a flow rate as high as 60 L/minute and a fraction of inspired oxygen (FiO2) of 0.21 to 1.0. An increased flow rate can help reduce inspiratory effort and improve dynamic lung compliance, which can lead to improving oxygenation and ventilation.1,5,6 During the COVID-19 pandemic, several studies showed that the use of HFNC as an alternative oxygen device for respiratory failure patients can provide higher oxygen concentrations than can be achieved with conventional devices, reduce the need for endotracheal intubation, and decrease the length of stay in intensive care units (ICUs).7–9 In addition, HFNC has a valuable effect in reducing anatomical dead space, which provides low positive end-expiratory pressure (PEEP) and can be more tolerable for patients with chronic obstructive pulmonary disease (COPD).10 Moreover, the immediate use of HFNC for postoperative respiratory failure patients was associated with lower risks of reintubation and respiratory failure.11
Even though the use of HFNC is prevalent in clinical settings, RTs still lack evidence-based guidelines for implementing HFNC.12 Moreover, HFNC is frequently utilized by RTs but controversy remains on the initiation, management and weaning of HFNC due to scant evidence.12–14 A cross-sectional study of French ICU physicians found that there was a great deal of variability in the current use of HFNC, including the criteria for initiation and weaning.15 Furthermore, a global survey of intensive care unit (ICU) healthcare providers reported that there was considerable variation in the daily application of HFNC with regard to initial settings and management criteria for HFNC parameters.16 HFNC failure may result from these notable differences in clinical practice, which is probably due to lack of educational training and standardized protocols.17
Despite the wide use of HFNC and the studies exploring the benefits of using HFNC with ARDS patients, research into assessing the knowledge, practice, and barriers to using HFNC among respiratory therapists in Saudi Arabia is lacking. Therefore, this study aimed to assess the current practice of HFNC in multiple centers in Saudi Arabia and identify the barriers to using HFNC among respiratory therapists.
Methods
Study Design
In this cross-sectional study, a survey was distributed through an electronic platform SurveyMonkey between December 19, 2022, and July 15, 2023.
Instrument
Experts in the use of HFNC (namely ICU physicians, respiratory therapists, and ICU nurses) formulated this questionnaire, which was adapted and modified from previous studies.8,15,16,18 Next, face and content validity were assessed by an expert panel and the survey was then pilot-tested. After receiving feedback from the experts, adjustments were made, and the survey was distributed. The survey consisted of six main parts:
- The first part asked the participants about their demographic information (eg, gender, number of years working in critical care areas, and geographical location).
- The second part focused on assessing the study participants’ knowledge of indications for the use of HFNC. This part consists of 13 statements with a five-point Likert scale ranging from 1 to 5 (1 = strongly agree; 5 = strongly disagree).
- The third part consisted of four statements with a five-point Likert scale ranging from 1 to 5 (1 = strongly agree; 5 = strongly disagree). This section assessed whether respiratory therapists agree that 1) the HFNC device is tolerable for patients, 2) HFNC is used to help patients eat and speak, 3) HFNC improves dyspnea, and 4) HFNC is used to avoid the need for intubation and invasive mechanical ventilation.
- The fourth part assessed the study participants’ ability to determine the timing for switching to HFNC, the most appropriate initial settings for HFNC, the management of patients on HFNC, and the criteria required for weaning.
- The fifth part consisted of four statements to evaluate whether the study participants were aware of the criteria for HFNC failure and the time to intubate and use invasive mechanical ventilation.
- The last part consisted of one question about barriers to HFNC use.
Data Collection and Sampling
A convenience sampling strategy was used to recruit the study participants, and the main target population for this study were respiratory therapists who work in Saudi Arabian hospitals and hold a bachelor degree. To reach a larger population of respiratory therapists, we distributed the survey through Saudi scientific non-profit organizations (eg, the Saudi Society for Respiratory Care) along with social media platforms (X and WhatsApp). In addition, each data collector visited and circulated the survey to respiratory therapists working at their nearby tertiary hospitals.
Data Analysis
The collected data were managed and analyzed using the Statistical Package for the Social Sciences (SPSS) version 27. Data were presented as means and standard deviation or frequency and percentages, as appropriate. A Chi-square test was used to compare RT staff who have existing HFNC protocols and those who do not. A p-value of <0.05 was considered statistically significant.
Ethical Approval
Before the commencement of the study, ethical approval was obtained from the Bioethical Committee at Batterjee Medical College (Reference Number RES-2022-0077).
Results
Participant Characteristics
In this study, 1001 RT practitioners completed the online survey. Over half of the participants were female (573, or 57.2%). The majority of our sample was staff specialists (824, or 82.3%), while directors of RT departments accounted for 29 (2.9%). High numbers of representatives were from the country’s Western and Central Regions (52.8% and 26.4%, respectively). More than half (56.1%) of the participants had one to five years of clinical experience. In addition, we documented the areas where the respiratory therapists were assigned, such as critical care (78.6%), non-critical care (65.5%), and ER (57.9%). Two-thirds of the respiratory therapists (659, or 65.8%) had received training on HFNC and 785 (78.4%) had used HFNC in clinical settings. (See Table 1).
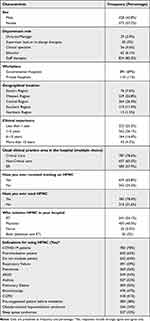 |
Table 1 Demographic Data and Characteristics of Study Respondents (n = 1001)
|
Indications of Using High-Flow Nasal Cannula from the Perspective of RT Staff
We asked the participants if they agreed about the relevant indications or conditions that require HFNC. The top responses were COVID-19 (78%), post-extubation (65%), and do-not-intubate patients (64%). The lowest responses were pre-oxygenated patients before intubation (38%), obesity-induced hypoventilation syndrome (34%), and sleep apnea syndrome (33%). (See Table 1 and Figure 1).
 |
Figure 1 Indications for using HFNC from the perspective of RT staff (n=1001).
|
Advantages of Using High-Flow Nasal Cannula
Four advantages of HFNC (ie, has high tolerability, helps maintain conversation and eating abilities, improves shortness of breath (SOB), and avoids intubation) were reported from the perspective of respiratory therapists. Participants strongly agreed that the advantages of HFNC were helping to maintain conversation and eating abilities (32.95%) and improving SBO (34.1%) (See Table 2 and Figure 2).
 |
Table 2 Advantages of Using HFNC from the Perspective of RT Staff
|
 |
Figure 2 Advantages of using HFNC from the prospective of RT staff (n=1001).
|
Initial Settings, Weaning and Disconnect of High-Flow Nasal Cannula
Surprisingly, 568 (57%) of RT staff do not follow a protocol for initiation, weaning and disconnection of HFNC with ARDS patients.
When starting HFNC, 40.2% of the participants started with FiO2 of 61% to 80%. Additionally, a high percentage of RT staff (40.6%) started with a flow rate between 30 L/minute and 40 L/minute and a temperature of 37°C (57.7%). When weaning ARDS patients from HFNC, 482 (48.1%) recommended first reducing gas flow by 5–10 L/minute every two to four hours, followed by 362 (36.2%) who recommended first reducing FiO2 by 5–10% every two to four hours. Moreover, 549 (54.8%) believed that ARDS patients could be disconnected from HFNC if they achieved a flow rate < 20 L/minute and FiO2 <35%. (See Table 3).
 |
Table 3 Clinical Practice of HFNC
|
In addition, we compared the responses between participants who followed a standardized protocol versus those who did not follow a protocol to initiate, wean and disconnect HFNC. (See Table 3).
Of the 402 RT staff who would start with FiO2 between 61% and 80%, 289 (28.87%) reported not following a standardized protocol. Additionally, half of the RT staff (214, or 21.42%) who would start with a flow rate between 30 and 40 L/minute do not have a standardized protocol to follow. Among all participants, we observed a statistically significant difference between respiratory therapists who follow a protocol versus those who do not (p-value <0.001). For those who recommended first reducing gas flow by 5–10 L/minute every two to four hours when weaning ARDS patients, 356 (35.51%) did not follow a standardized protocol. Among all participants, we observed statistically significant differences between respiratory therapists who follow a protocol and those who do not follow a protocol (p-value <0.001). (See Table 3).
Criteria to Stop High-Flow Nasal Cannula and Intubate Acute Respiratory Distress Syndrome Patients
Regarding the criteria to stop providing HFNC and initiate intubation, 39.5% of the participants would stop providing HFNC if the patient paused or stopped breathing. Indeed, 34.7% of the participants would stop providing HFNC in cases of refractory hypoxemia (SpO2 < 90% with FiO2 100% and flow rate of 60 L/minute), 35% in cases of severe respiratory acidosis (pH is unacceptably low (<7.25) and the PaCO2 is elevated), and 39.3% in cases of reduced level of consciousness (GCS score ≤8). (See Table 4).
 |
Table 4 Criteria to Stop HFNC and Intubate ARDS Patients
|
Challenges in Using High-Flow Nasal Cannula
Regarding the challenges that impede the use of HFNC, participants ranked lack of knowledge, lack of devices, and the absence of protocol as the highest challenges (57.3%, 49.6, and 49%, respectively) while lack of evidence and diversity of HFNC devices were the lowest challenges 34.5% and 17.2%, respectively. (See Figure 3).
 |
Figure 3 Challenges toward using HFNC from the perspective of RT staff (n=1001).
|
Discussion
To the best of our knowledge, this is the first national study to shed light on the knowledge, attitudes, and current practice of HFNC in ARDS patients among respiratory therapists in Saudi Arabia and the barriers to its use in clinical settings. The study findings revealed nuanced applications marked by significant endorsement in certain clinical scenarios and a lack of protocol adherence, underscoring the need for uniform, evidence-based guidelines and enhanced training for respiratory therapists.
A significant finding in our study is the prominent endorsement of HFNC use in COVID-19 patients, post-extubation cases, and do-not-intubate patients. These results corroborate existing literature that underscores HFNC’s role in enhancing oxygenation and reducing the effort of breathing in acute hypoxemic respiratory failure.2,3 Similarly, a cross-sectional study disclosed that respiratory physicians in Japan regarded COVID-19 as a primary indicator for HFNC application given its propensity to reduce the frequency of sustained sedation, physical restraint, and length of stay in the ICU compared to patients undergoing non-invasive ventilation (NIV).8 Nevertheless, the lower agreement on HFNC’s application in pre-oxygenation, obesity-induced hypoventilation, and sleep apnea conditions indicates potential knowledge gaps or diverse clinical experiences that warrant further investigation.
HFNC therapy has garnered widespread clinical validation for its efficacy in the management of patients with ARDS. Its capacity to deliver a precise and adjustable flow of warmed, humidified oxygen optimizes patient comfort and oxygenation status and decreases respiratory distress symptoms and the risk of endotracheal intubation.19,20 Within the scope of the present study, a substantial level of consensus was observed among participants, indicating that HFNC application is associated with a notable amelioration of SOB while concurrently preserving speech and eating capabilities. In line with these findings, previous literature has demonstrated the superiority of HFNC over alternative non-invasive respiratory modalities, underscored by its high patient tolerance and preservation of patients’ daily activities, emphasizing HFNC’s pivotal role in enhancing patient-centered outcomes.8
In the current study, significant discrepancies were observed in the setting of initial parameters, weaning strategies, and disconnection criteria pertaining to HFNC. Specifically, a majority of respiratory therapists reported an initial application of FiO2 within the range of 60% to 80%, a flow rate of 30–40 L/minute, and a temperature of 37°C. Notably, these findings are inconsistent with established HFNC protocols, which advocate initiating FiO2 at 1.00, a flow rate of 60 L/minute, and a temperature of 37°C.21,22 Furthermore, a substantial proportion of respiratory therapists expressed a preference for initially weaning the flow rate by 5–10 L/minute every two to four hours, deviating from the guidelines stipulated by the Canadian Society of Respiratory Therapists, which prioritize reducing FiO2 to less than 40% before commencing a gradual decrement in flow rate by increments of five.21 Analogously, heterogeneous practices were observed among French ICU physicians during the weaning process of HFNC settings.15 Despite these variations, a significant consensus emerged among over half of the respiratory therapists regarding the disconnection of HFNC, advocating for disconnection when the FiO2 is below 35% and the flow rate falls below 20 L/minute, aligning with published guidelines.1,21 A noteworthy revelation is the lack of adherence to standardized protocols for initiating, weaning, and disconnecting HFNC for ARDS patients. This inconsistency in clinical practices underscores the urgent need for the development and dissemination of evidence-based guidelines to enhance the quality and consistency of patient care.
Regarding the criteria dictating the transition from HFNC therapy to invasive mechanical ventilation in patients with ARDS, our study revealed a moderate consensus. This alignment was substantiated through the initiation of endotracheal intubation in cases of spontaneous breathing cessation, refractory hypoxemia, severe respiratory acidosis, or diminished consciousness. Concomitantly, a subsequent study yielded analogous outcomes, thereby affirming our findings. Notably, it illuminated the exacerbation of respiratory distress and the presence of bronchial congestion as substantive contributors to the ineffectiveness of HFNC therapy, consequently augmenting the imperative for invasive ventilation.15 Additionally, a retrospective analysis furnished empirical evidence identifying hypoxemic and hypercapnic respiratory failure as crucial indicators denoting the failure of HFNC therapy, particularly in scenarios wherein patients cannot sustain SpO2 above 90% despite receiving maximal FiO2. This insufficiency is accentuated by concomitant findings of arterial pH below 7.3 and respiratory rates exceeding 35 breaths per minute.23 The diverse criteria employed by respiratory therapists to discontinue HFNC and commence intubation emphasize the need for standardization in this aspect. Precisely articulated and evidence-based criteria are instrumental in optimizing patient outcomes and mitigating the potential risks associated with delayed intubation.
Several barriers impede the widespread adoption of HFNC therapy in clinical settings. In the present study, RT staff identified a lack of knowledge, the unavailability of devices, and the absence of protocols as the foremost challenges hindering the optimal use of HFNC. In accordance with these results, existing literature has shown that a lack of skill and familiarity hampers the implementation of HFNC in the ICU.24,25 Likewise, a pertinent study revealed that the absence of established objective criteria for initiating and managing HFNC settings significantly restricts its application, potentially leading to ineffective healthcare practices.26 These findings underscore the necessity for targeted interventions to enhance the capabilities of respiratory therapists, ensure the availability of essential equipment, and establish evidence-based protocols and the importance of concerted efforts to fortify strengths and mitigate the challenges associated with HFNC application in clinical settings.
Strengths and Limitations
One of the notable strengths of this study is the breadth of the participant sample, offering a robust spectrum of insights and experiences from a diverse group of respiratory therapists. The comprehensive survey methodology enabled the capture of nuanced, multifaceted data, allowing for a granular analysis of HFNC practices and challenges. Additionally, the alignment of the study’s findings with existing literature accentuates its validity and contribution to the ongoing discourse on the clinical applications of HFNC. Nevertheless, the study has limitations. We did not assess the current practice and barriers of using HFNC from the prospective of physicians whose practices maybe different from RTs. The reliance on self-reported data introduces the potential for response biases, wherein participants might either consciously or unconsciously misreport their practices. The cross-sectional nature of the study design precludes causal inferences and the assessment of HFNC practice evolution over time. Moreover, the geographical and institutional diversity of participants, while a strength, also raises questions about the generalizability of the findings across different contexts and healthcare systems.
Conclusion
Respiratory therapists in Saudi Arabia demonstrated a profound understanding of the clinical advantages associated with the utilization of HFNC for adult patients with ARDS. However, significant discrepancies were observed concerning the setting of initial parameters, the formulation of weaning strategies, and the determination of disconnection criteria related to HFNC. These variations primarily stemmed from inadequate adherence to established protocols and limitations in available resources. Such observations underscore the imperative for the development and implementation of standardized, evidence-based guidelines, alongside comprehensive training initiatives aimed at enhancing respiratory therapists’ compliance with HFNC protocols.
Data Sharing Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Ethical Approval
This study was approved from Bioethical Committee at Batterjee Medical College (Reference Number RES-2022-0077) and conducted in accordance with the Declaration of Helsinki. Informed consent was obtained from all RTs participated in the study.
Author Contributions
All authors made a significant contribution to the work reported, whether that is in the conception, study design, execution, acquisition of data, analysis and interpretation, or in all these areas; took part in drafting, revising or critically reviewing the article; gave final approval of the version to be published; have agreed on the journal to which the article has been submitted; and agree to be accountable for all aspects of the work.
Funding
There is no financial supporting body for this study.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Park S. High-flow nasal cannula for respiratory failure in adult patients. Acute Crit Care. 2021;36(4):275–285. doi:10.4266/acc.2021.01571
2. Qaseem A, Etxeandia-Ikobaltzeta I, Fitterman N, Williams JW Jr, Kansagara D; Physicians CGCotACo. Appropriate use of high-flow nasal oxygen in hospitalized patients for initial or postextubation management of acute respiratory failure: a clinical guideline from the American College of Physicians. Ann Internal Med. 2021;174(7):977–984. doi:10.7326/M20-7533
3. Papazian L, Corley A, Hess D, et al. Use of high-flow nasal cannula oxygenation in ICU adults: a narrative review. Intensive Care Med. 2016;42(9):1336–1349. doi:10.1007/s00134-016-4277-8
4. Nishimura M. High-flow nasal cannula oxygen therapy devices. Respir Care. 2019;64(6):735–742. doi:10.4187/respcare.06718
5. Mauri T, Alban L, Turrini C, et al. Optimum support by high-flow nasal cannula in acute hypoxemic respiratory failure: effects of increasing flow rates. Intensive Care Med. 2017;43(10):1453–1463. doi:10.1007/s00134-017-4890-1
6. Mauri T, Turrini C, Eronia N, et al. Physiologic effects of high-flow nasal cannula in acute hypoxemic respiratory failure. Am J Respir Crit Care Med. 2017;195(9):1207–1215. doi:10.1164/rccm.201605-0916OC
7. Gürün A, Öz M, Erol S, Çiftçi F, Çiledağ A, Kaya A. High flow nasal cannula in COVID-19: a literature review. Tuberk Toraks. 2020;68(2):168–174. doi:10.5578/tt.69807
8. Koyauchi T, Suzuki Y, Inoue Y, et al. Clinical practice of high-flow nasal cannula therapy in COVID-19 pandemic era: a cross-sectional survey of respiratory physicians. Respir Invest. 2022;60(6):779–786. doi:10.1016/j.resinv.2022.08.007
9. Sayan İ, Altınay M, Çınar AS, et al. Impact of HFNC application on mortality and intensive care length of stay in acute respiratory failure secondary to COVID-19 pneumonia. Heart Lung. 2021;50(3):425–429. doi:10.1016/j.hrtlng.2021.02.009
10. Millar J, Lutton S, O’Connor P. The use of high-flow nasal oxygen therapy in the management of hypercarbic respiratory failure. Ther Adv Respir Dis. 2014;8(2):63–64. doi:10.1177/1753465814521890
11. Hernández G, Vaquero C, González P, et al. Effect of postextubation high-flow nasal cannula vs conventional oxygen therapy on reintubation in low-risk patients: a randomized clinical trial. JAMA. 2016;315(13):1354–1361. doi:10.1001/jama.2016.2711
12. Alnajada A, Blackwood B, Messer B, Pavlov I, Shyamsundar M. International survey of high-flow nasal therapy use for respiratory failure in adult patients. J Clin Med. 2023;12(12):3911. doi:10.3390/jcm12123911
13. Puah SH, Li A, Cove ME, et al. High-flow nasal cannula therapy: a multicentred survey of the practices among physicians and respiratory therapists in Singapore. Aust Crit Care. 2022;35(5):520–526. doi:10.1016/j.aucc.2021.08.001
14. Alnajada A, Shyamsundar M, Messer B, Pavlov I. North American Survey of High-Flow Nasal Cannula Therapy Use. A42 ARF/ARDS. American Thoracic Society; 2022:A5535–A5535.
15. Besnier E, Hobeika S, NSeir S, et al. High-flow nasal cannula therapy: clinical practice in intensive care units. Ann Intens Care. 2019;9:1–8.
16. Li J, Tu M, Yang L, et al. Worldwide clinical practice of high-flow nasal cannula and concomitant aerosol therapy in the adult ICU setting. Respir Care. 2021;66(9):1416–1424. doi:10.4187/respcare.08996
17. Wen R, Hu X, Wei T, et al. High-flow nasal cannula: evaluation of the perceptions of various performance aspects among Chinese clinical staff and establishment of a multidimensional clinical evaluation system. Front Med. 2022;9:900958. doi:10.3389/fmed.2022.900958
18. Hosheh O, Edwards CT, Ramnarayan P. A nationwide survey on the use of heated humidified high flow oxygen therapy on the paediatric wards in the UK: current practice and research priorities. BMC Pediatr. 2020;20(1):109. doi:10.1186/s12887-020-1998-1
19. Abdelbaky AM, Elmasry WG, Awad AH, Khan S, Jarrahi M. The impact of high-flow nasal cannula therapy on acute respiratory distress syndrome patients: a systematic review. Cureus. 2023;15(6):1.
20. Lee CC, Mankodi D, Shaharyar S, et al. High flow nasal cannula versus conventional oxygen therapy and non-invasive ventilation in adults with acute hypoxemic respiratory failure: a systematic review. Respir Med. 2016;121:100–108. doi:10.1016/j.rmed.2016.11.004
21. Ischaki E, Pantazopoulos I, Zakynthinos S. Nasal high flow therapy: a novel treatment rather than a more expensive oxygen device. Eur Respir Rev. 2017;26(145):170028. doi:10.1183/16000617.0028-2017
22. Vianello A, Arcaro G, Molena B, et al. High-flow nasal cannula oxygen therapy to treat patients with hypoxemic acute respiratory failure consequent to SARS-CoV-2 infection. Thorax. 2020;75(11):998–1000. doi:10.1136/thoraxjnl-2020-214993
23. Kang BJ, Koh Y, Lim C-M, et al. Failure of high-flow nasal cannula therapy may delay intubation and increase mortality. Intensive Care Med. 2015;41(4):623–632. doi:10.1007/s00134-015-3693-5
24. O’Brien SL, Haskell L, Tavender EJ, et al. Factors influencing health professionals’ use of high-flow nasal cannula therapy for infants with bronchiolitis–A qualitative study. Front Pediatr. 2023;11:1098577. doi:10.3389/fped.2023.1098577
25. Jackson JA, Spilman SK, Kingery LK, et al. Implementation of high-flow nasal cannula therapy outside the intensive care setting. Respir Care. 2021;66(3):357–365. doi:10.4187/respcare.07960
26. Horvat CM, Pelletier JH. High-flow nasal cannula use and patient-centered outcomes for pediatric bronchiolitis. JAMA Network Open. 2021;4(10):e2130927–e2130927. doi:10.1001/jamanetworkopen.2021.30927