Pentru Trendy zircon o-z de aur alfabetul aliaj străpuns de unghii de arta farmece 26 de litere manichiura decoratiuni accesorii en-gros de dropshipping > Nails Art & Instrumente / Labordiagnosticatm.ro

Set cu unghii false Beauty Engros | Jucarii infrumusetare Engros | Jucarii Engros | Articole pentru copii Engros

Set de unghii false si accesorii pentru fetite, multicolor, 16×14.5cm, +3ani, en-gros | Jucarii infrumusetare Engros | Jucarii Engros | Articole pentru copii Engros

60 buc. Forme duble Extensie pentru unghii false Crystal Clear Sfaturi pentru unghii cu acoperire completa Deget fals Accesorii profesionale pentru manichiura Instrumente - Baduglobal.ro

Ușor de purtat, Nail Art Alb Stele Pearl Unghii False Lucios Scurt Sicriu Terminat Unghii False Apăsați pe Unghii cu Lipici en-Gros cumpara > Frumusețe & sănătate < Bonlavie.ro

Cumpără 24 buc/pungă Unghii false Apăsați pe set de sfaturi scurte reutilizabile, roz artificial, nud, sfaturi pentru unghii false Unghii lipite Unghiile pentru unghii DIY | Joom

Marmura roz pătrat unghii false lucios scurt model de piatra doamnelor falsies en-gros 24 de unghii false - Instrumente De Unghii / www.spau.ro

Hot pink rose mat unghii false trandafir rosu mat unghii false pătrat de top full nail art sfaturi apăsați pe deget accesorii manichiura - Instrumente De Unghii / www.spau.ro

Vanzare 20BUC/Set Lung Solid-culoare Unghii False Patch Mat ușor de Purtat Manichiura Accesorii Detașabile Nails Art Patch ~ Nails Art & Instrumente / www.andreimuresanusud.ro

24buc artificiale unghii false tips detasabila timp de balerina unghii false cu design portabil unghii false complet acoperi sfaturi de unghii vanzare < Instrumente De Unghii \ Bivoli.ro

Cumpără Fototerapie Patch pentru unghii Gel pentru manichiură Lipici pentru unghii false Sfaturi Instrument Fără deteriorare a unghiilor Lipici pentru unghii false Accesorii pentru unghii | Joom

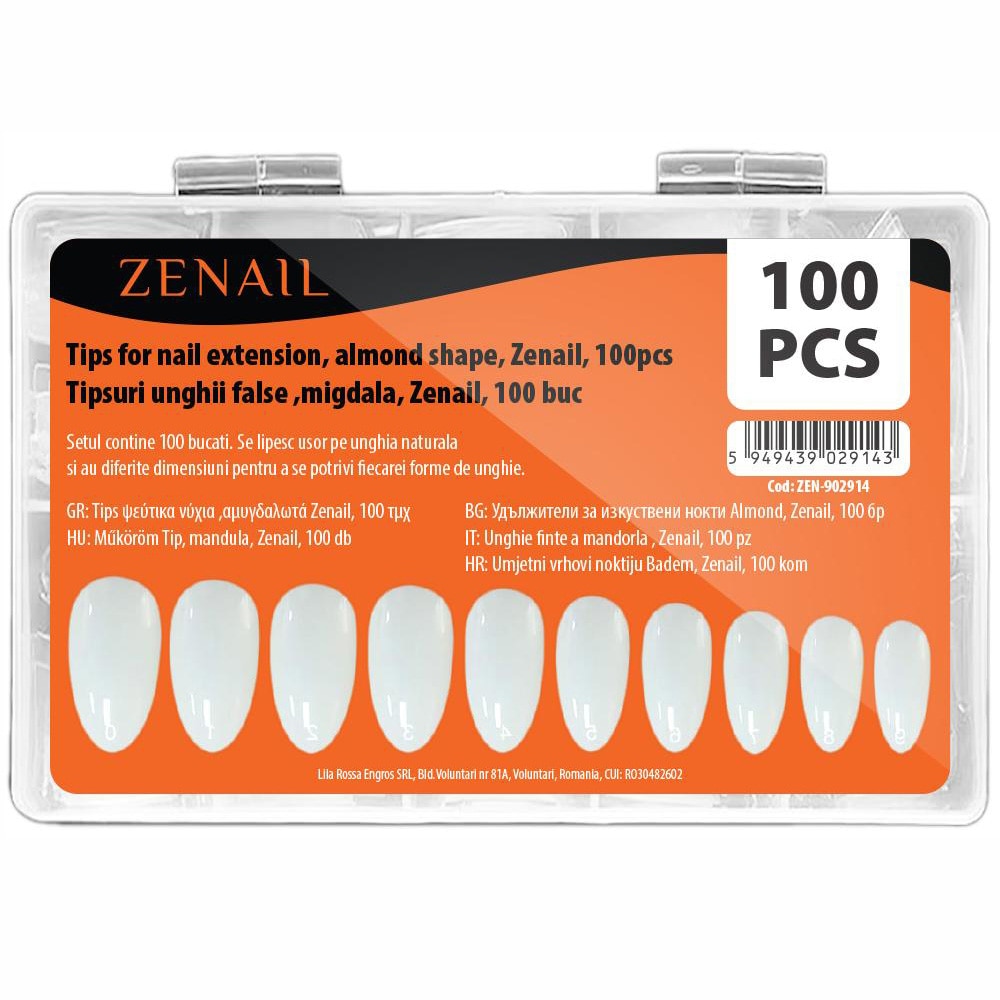
Produse unghii false | 100 tipsuri transparente pt Polygel | Produse unghii false la preturi de engros

3xl extra lungi stiletto acrilice apăsați pe unghii false sfaturi clare artificiale acoperi complet false degetul sculptat de unghii manichiura instrumente vanzare < Instrumente De Unghii \ Bivoli.ro

Forme duble din plastic pentru unghii Sfaturi false de artă pentru unghii pentru extensie de gel Forme acrilice pentru unghii Accesorii de stilizare profesionale Instrumente de manichiură - Baduglobal.ro