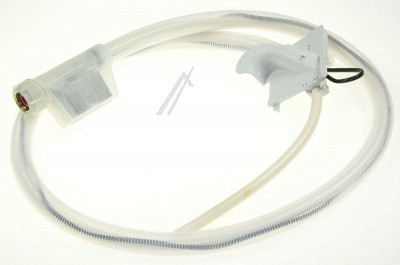
Bosch Mosogatógép alkatrész, Kifolyótömlő Bosch SGS3039 mosogatógéphez ew04073 háztartási gép kiegészítő vásárlás, olcsó Bosch Mosogatógép alkatrész, Kifolyótömlő Bosch SGS3039 mosogatógéphez ew04073 árak, akciók

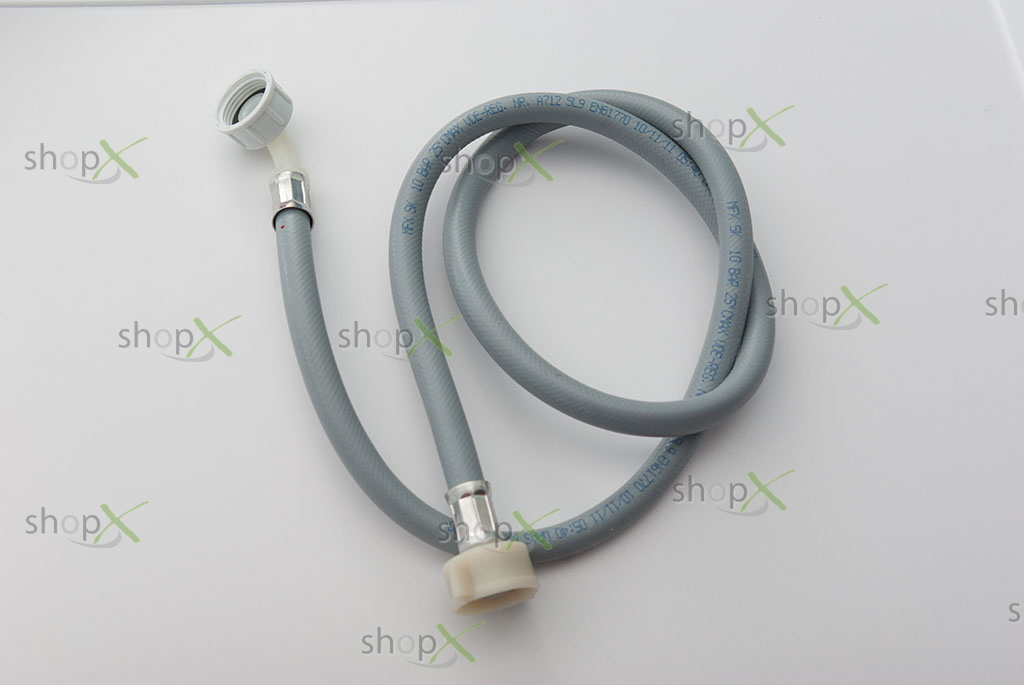
Mosogatógép befolyó cső - Gorenje alkatrészek - - Web shop - Teka Gorenje háztartási gép szerviz és alkatrész kereskedés

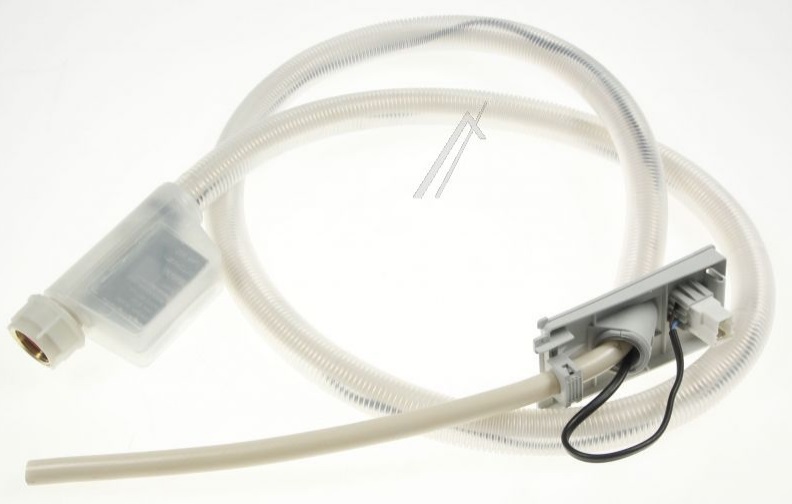
Mosógép, mosogatógép alkatrész AQUASTOP-BEFOLYÓTÖMLŐ 1,8M mosógéphez, mosogatógéphez ew03208 -Elektroweb Víztisztító, Háztartásigép alkatrészek - webáruház, webshop

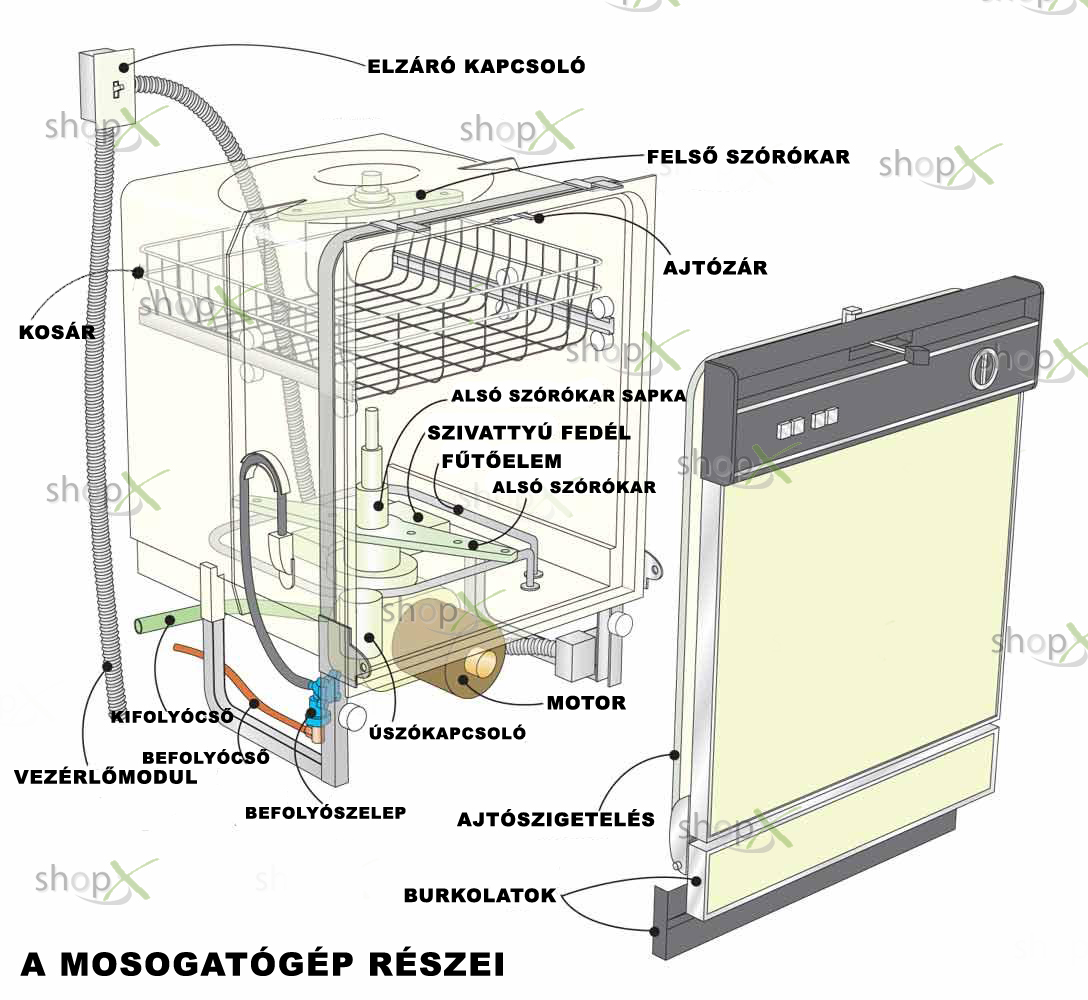
Bosch SGS2HVI20E Szabadonálló mosogatógép » 60 cm széles mosogatógép » Vásárlás » Bosch Siemens Márkabolt