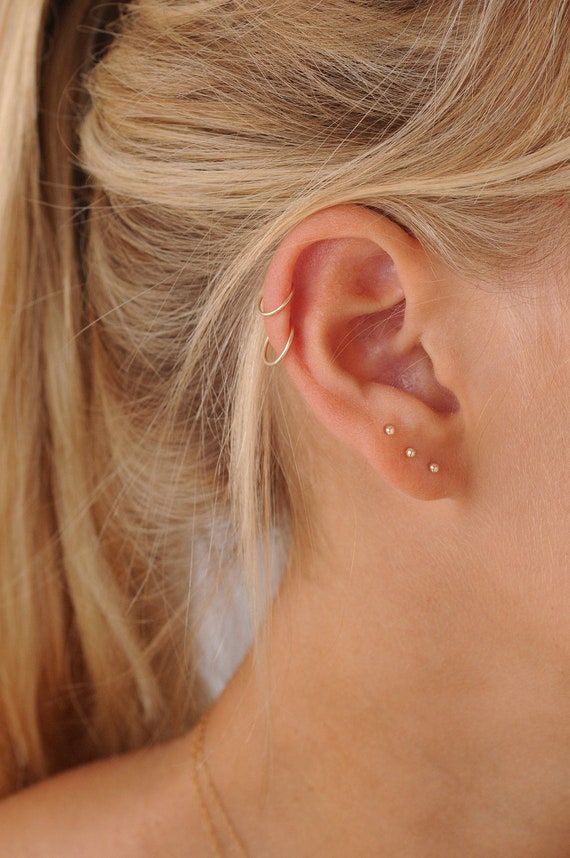
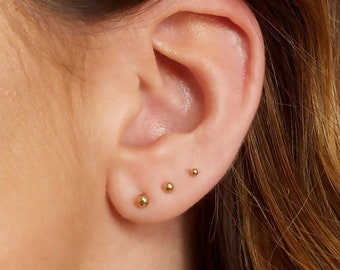
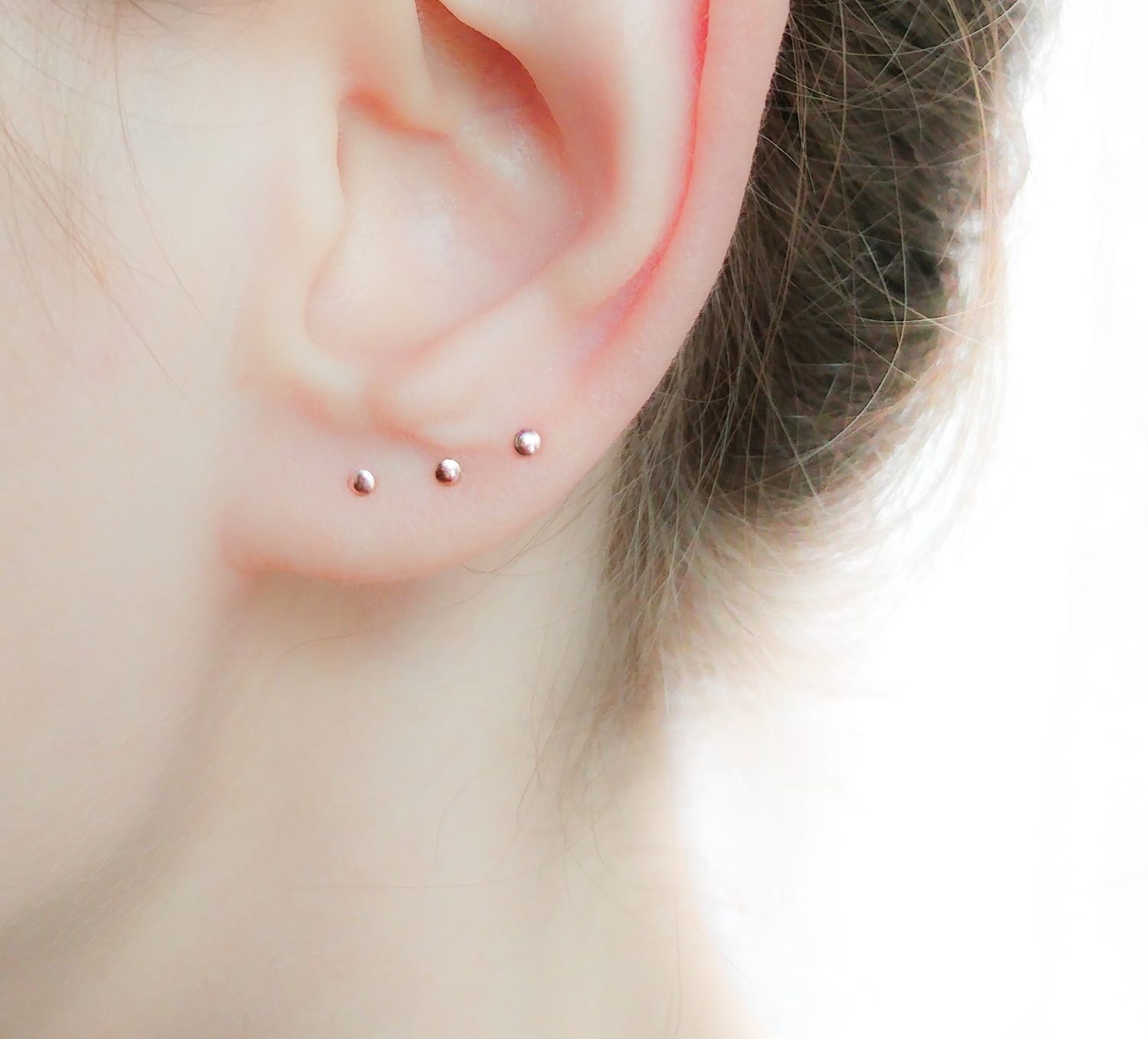
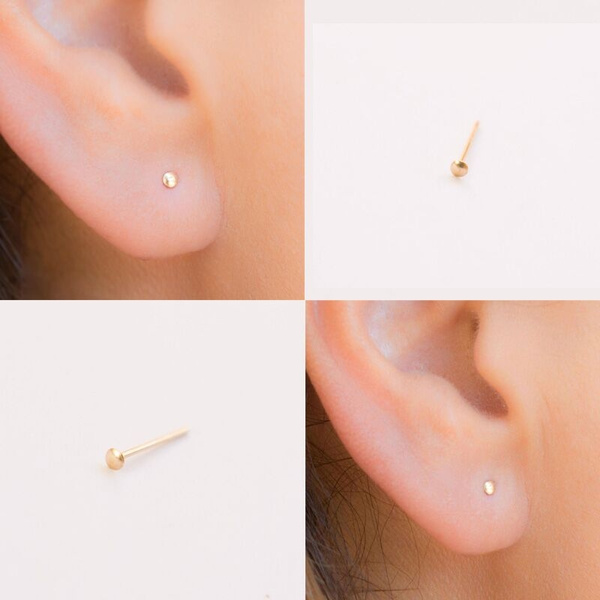
Tiny Gold Dot Studs Very Small Gold Stud Earrings 9ct Gold | Etsy UK | Earings piercings, Cool ear piercings, Cute ear piercings

Amazon.com: Tiny Earring Set, Gold Stud earrings, Small Bird Post Earring, 14K Solid Gold Studs, Unique Gold Earrings, Handmade Designer Jewelry : Handmade Products

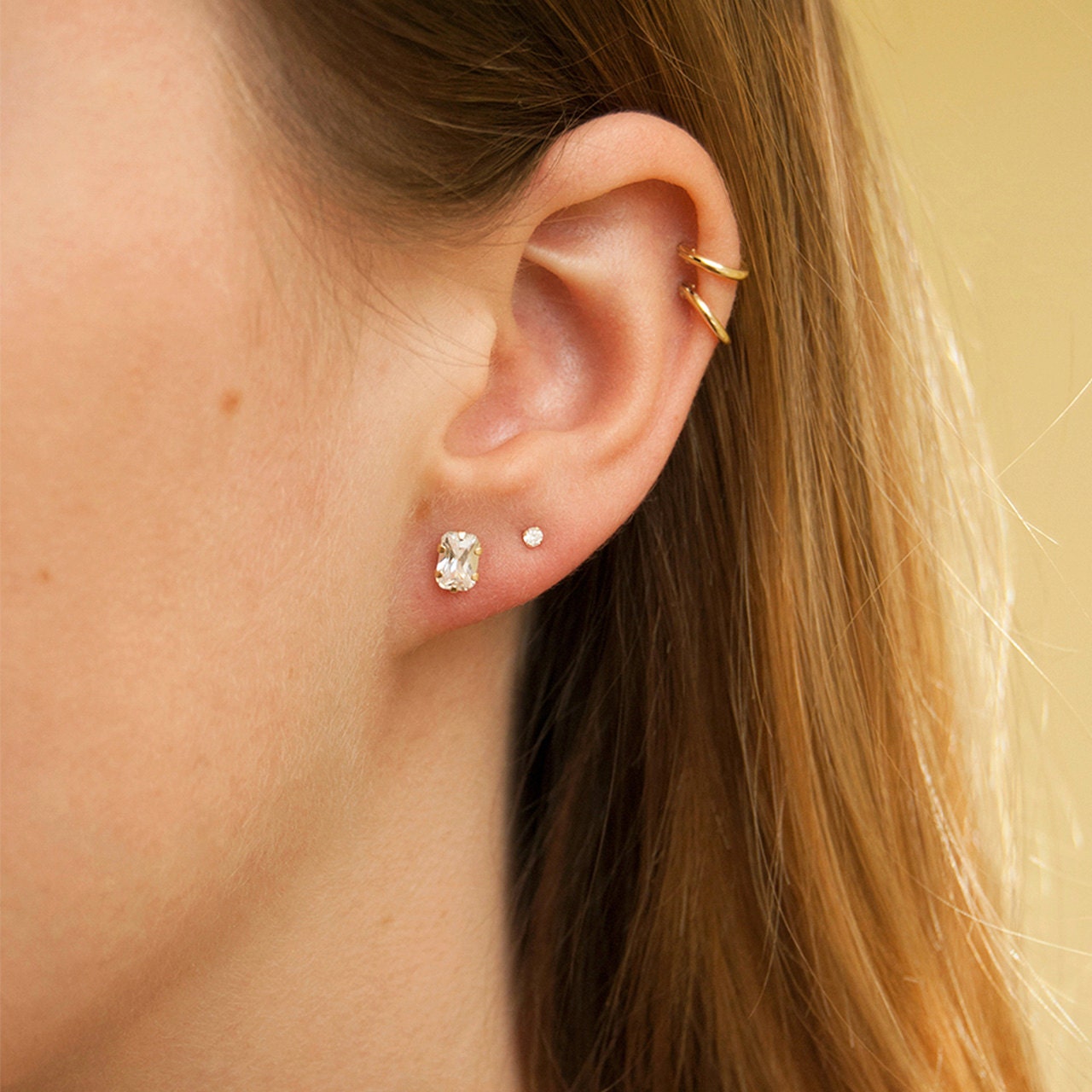
Gold Studs Earrings 14K Gold Filled Minimalist Women Studs | Etsy | Gold earrings studs simple, Simple gold earrings, Gold earrings studs
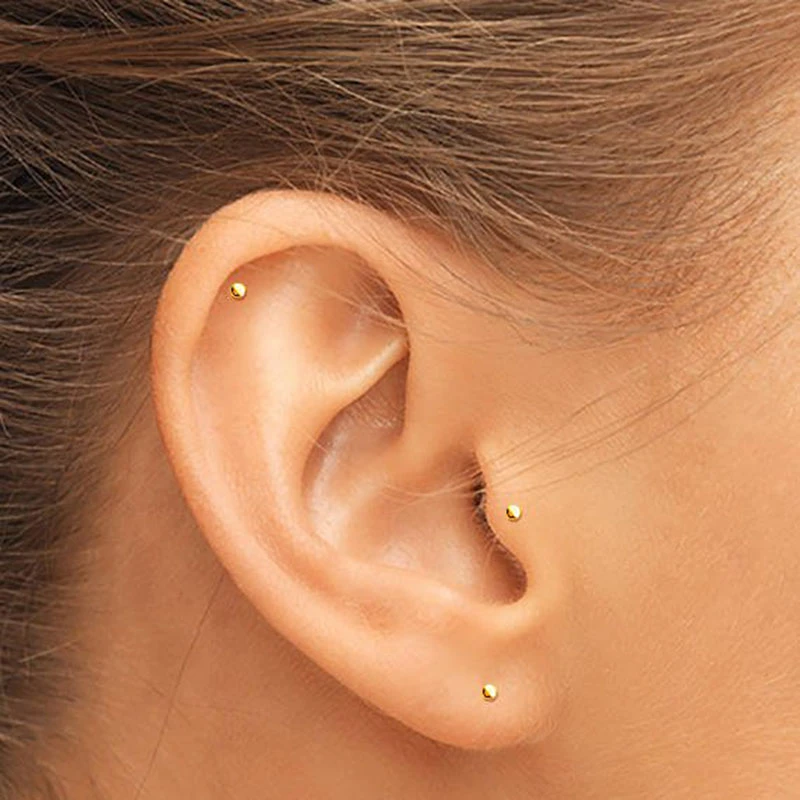
Domino Handmade Invisble Super small Gold Filled Tragus Stud Gold Stud Tiny Gold Studs Small Gold Earrings Simple Unique|Stud Earrings| - AliExpress

14k Solid Gold 4.7mm CZ Bezel Set Stud Earrings, Simple Gold Stud Earrings, Small Gold Stud Set #2944651 - Weddbook