20 قطعة/مجموعة من فرش رسم طلاء الأظافر مجموعة تصميم فرشاة تلميع قلم رسم ونقاط الرسم على الأظافر أدوات رسم الأظافر : Amazon.ae: الجمال

تخفيض صالون الأظافر 3 قطعة مسمار الفن قلم رصاص قلم رسم التنقيط أدوات عدة الأبيض غرامة طلاء أظافر بلاستيكية اللوحة ستريبينغ فرشاة SATR29 > أدوات الأظافر / Supermarket-Fast.cam

شراء أدوات الأظافر فرشاة الأظافر التنقيط لوحة رسم القلم مسمار الفن فرشاة جل البولندية فرش أدوات جل الطلاء القلم رخيص | التسليم السريع والجودة | Ar.Dhgate

الأظافر جمال العرض الاكريليك صندوق عناية بالأظافر مايكرو أدوات الرسم تدريبات الأظافر بالجملة - Buy أدوات الرسم الصغيرة ، تدريبات الأظافر بالجملة ، صندوق عناية بالأظافر من الأكريليك Product on Alibaba.com

شراء 15 قطعة / المجموعة المهنية Uv Gel Nail Art فرش مجموعة تصميم مسمار البولندية الطلاء رسم القلم مانيكير أدوات الأظافر رخيص | التسليم السريع والجودة | Ar.Dhgate

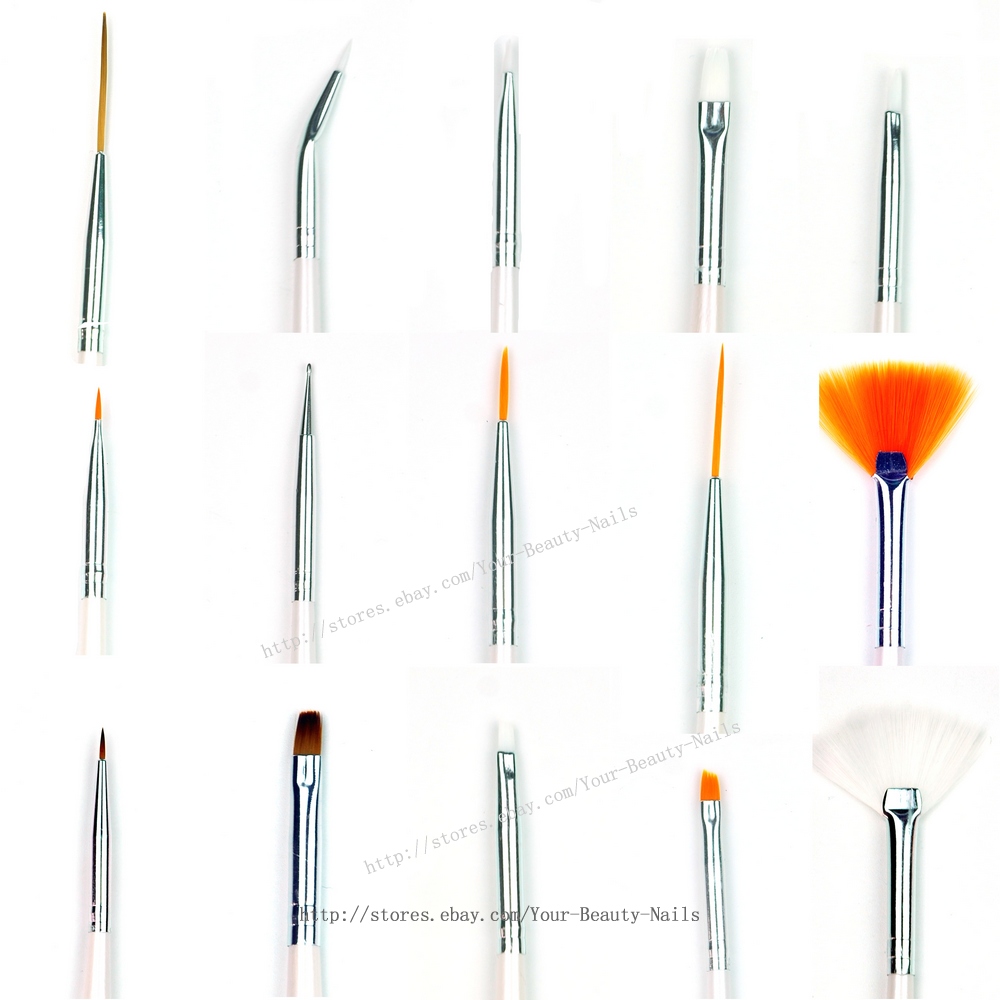
طقم رسم فني للأظافر التنقيط فرشاة الرسم البولندية أدوات القلم فرش الأظافر لأدوات الرسم الجل بالأشعة فوق البنفسجية 20 قطعة/المجموعة|nail brush|nail design brushespolishing brush - AliExpress

شراء المزدوج العضوية فرشاة الأظافر مشعر شقة مروج المخدرات التنقيط الأظافر نصائح اثنين من طرق استخدام مانيكير الأشعة فوق البنفسجية هلام البولندية بناء رسم الأظافر أدوات القلم رخيص | التسليم السريع والجودة

5 قطعة فرشاة الأظافر UV فرشاة جل تمديد اينر فرشاة الطلاء خطوط اينر رسم الأظافر القلم مانيكير مسمار أدوات الرسم / مسمار الفن و أدوات

شراء 7/9/11 / خفض 15mm فن الأظافر اينر فرشاة الرسم أدوات الرسم زهرة خطوط الفرنسية الشبكة الشريط الاكريليك الأشعة فوق البنفسجية هلام القلم DIY مانيكير JK1912 رخيص | التسليم السريع والجودة | Ar.Dhgate

قلم تحديد العيون للرسم على الأظافر ، قلم تحديد ، أدوات تخطيط ، طلاء أظافر ، خطاف ، تصميم منحوت على شكل زهرة / مسمار الفن و أدوات

نرسم على الأظافر للمبتدئين مع طلاء جل ، ألوان مائية ، دهانات أكريليك ، حرف واحد فقط. مخططات ، تعليمات للمبتدئين

Nail Art Tools and Sets Nail Brush Dotting Painting Drawing Pen Nail Polish Device For Manicure Nail Design|nail brush dot|dot paintingnail brush - AliExpress

فن الرسم على الاظافر Nail Art Drawing - ادوات الرسم على الاظافر ادوات تزين الاظافر 💟 #nailart #polish #nailpolish #nail #fashion #style #girl #beauty #glitter #sparkles #sparkle #art #الرسم_على_الاظافر #الاظافر #مناكير #

أدوات تحديد الأظافر/أقلام رسم Manicure/قلم رسم فني Nail - الصين صور الأظافر نملة، قلم حبر كروي، قلم لتلميع نويل، أقلام فنية لراميل، قلم فني ثلاثي الأبعاد، قلم طابعة ثلاثي الأبعاد، لؤلؤ صغير،

24 قطعة ليوبارد أظافر صناعية طويلة الفرنسية وهمية الأظافر اضغط على الأظافر لهب نمط مسمار نصائح مع الغراء مسمار أدوات الرسم | False Nails | Naoko

3 قطعة Uv جل مانيكير فرشاة اينر اللوحة القلم رسم نحت القلم أظافر تركيب من الأكريليك خط نصائح فرش Pensel مسمار أدوات الرسم القلم خصم \ أدوات الأظافر ~ Showroom-Money.cam