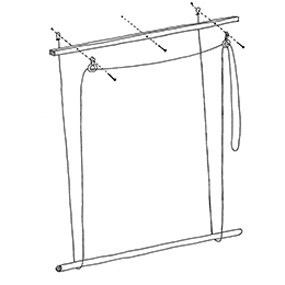
Heligo Veckat Klemmfix plisserad rullgardin utan borrning vit 35 x 80 cm (B x H) sekretess-solpersienn klämfix rullgardiner för fönster och dörr, lätt genomtränglig vikbar rullgardin rullgardin med klämfix-hållare : Amazon.se: Hem

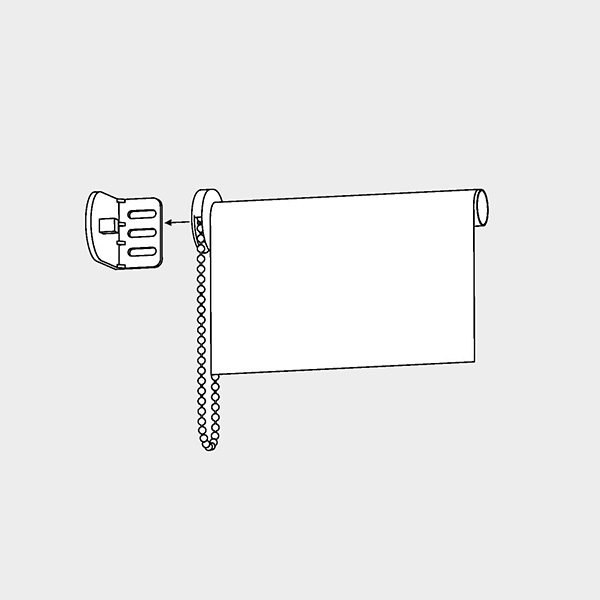
Fodlon 1 set rullgardin hållare fönster 28 mm rullgardin klämfäste reservdel, plast rullgardin beslag ersättningsset kedjetåg med skruvar för klämfix-persienner, dubbelrulle, vit : Amazon.se: Hem & kök

Fodlon 1 set rullgardin hållare fönster 28 mm rullgardin klämfäste reservdel, plast rullgardin beslag ersättningsset kedjetåg med skruvar för klämfix-persienner, dubbelrulle, vit : Amazon.se: Hem & kök