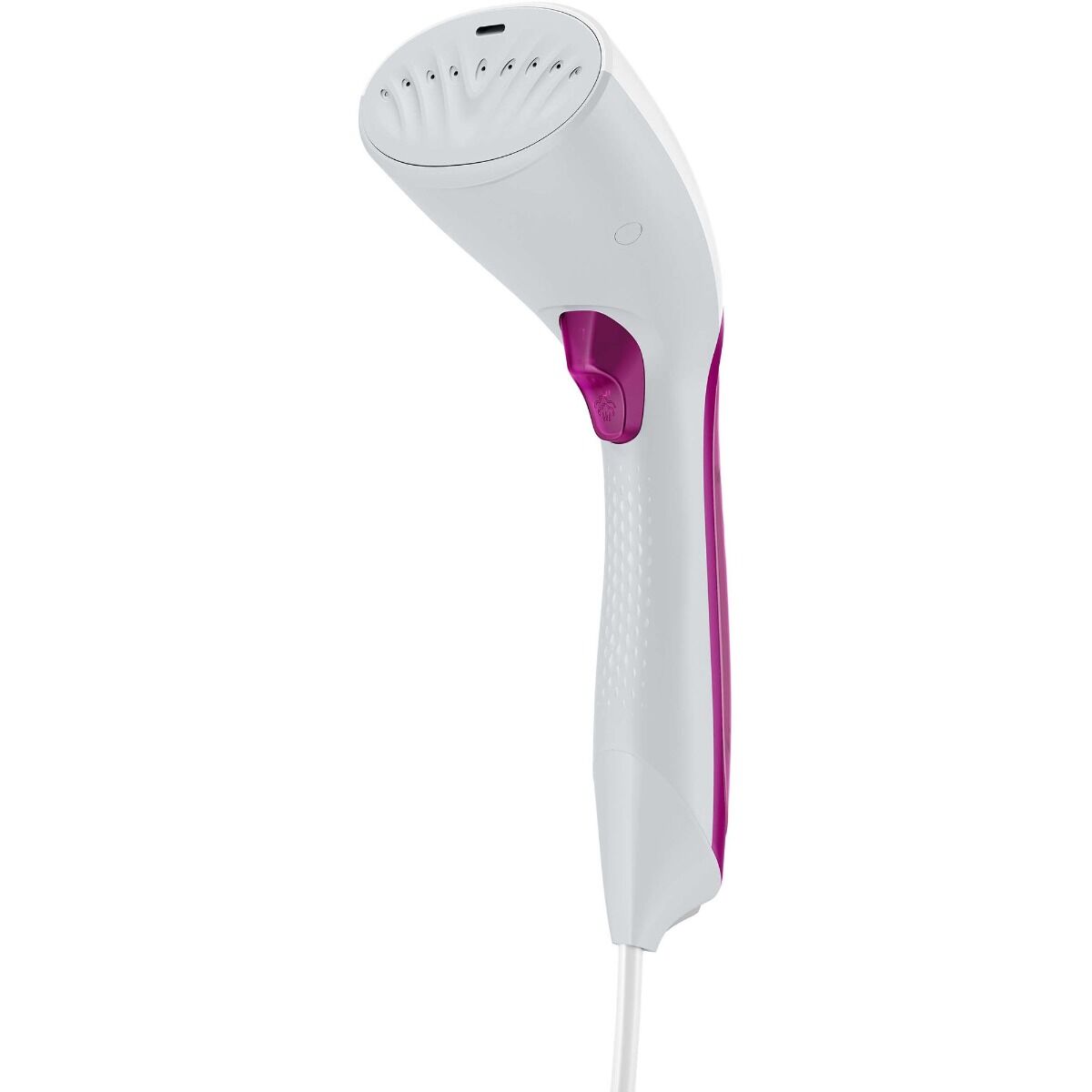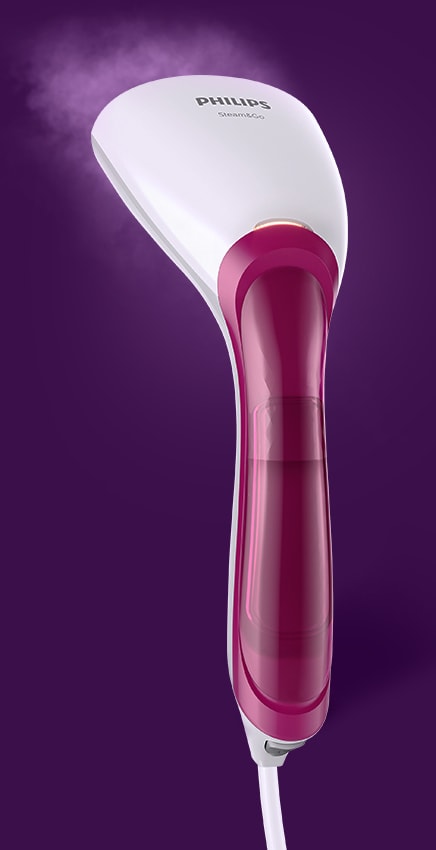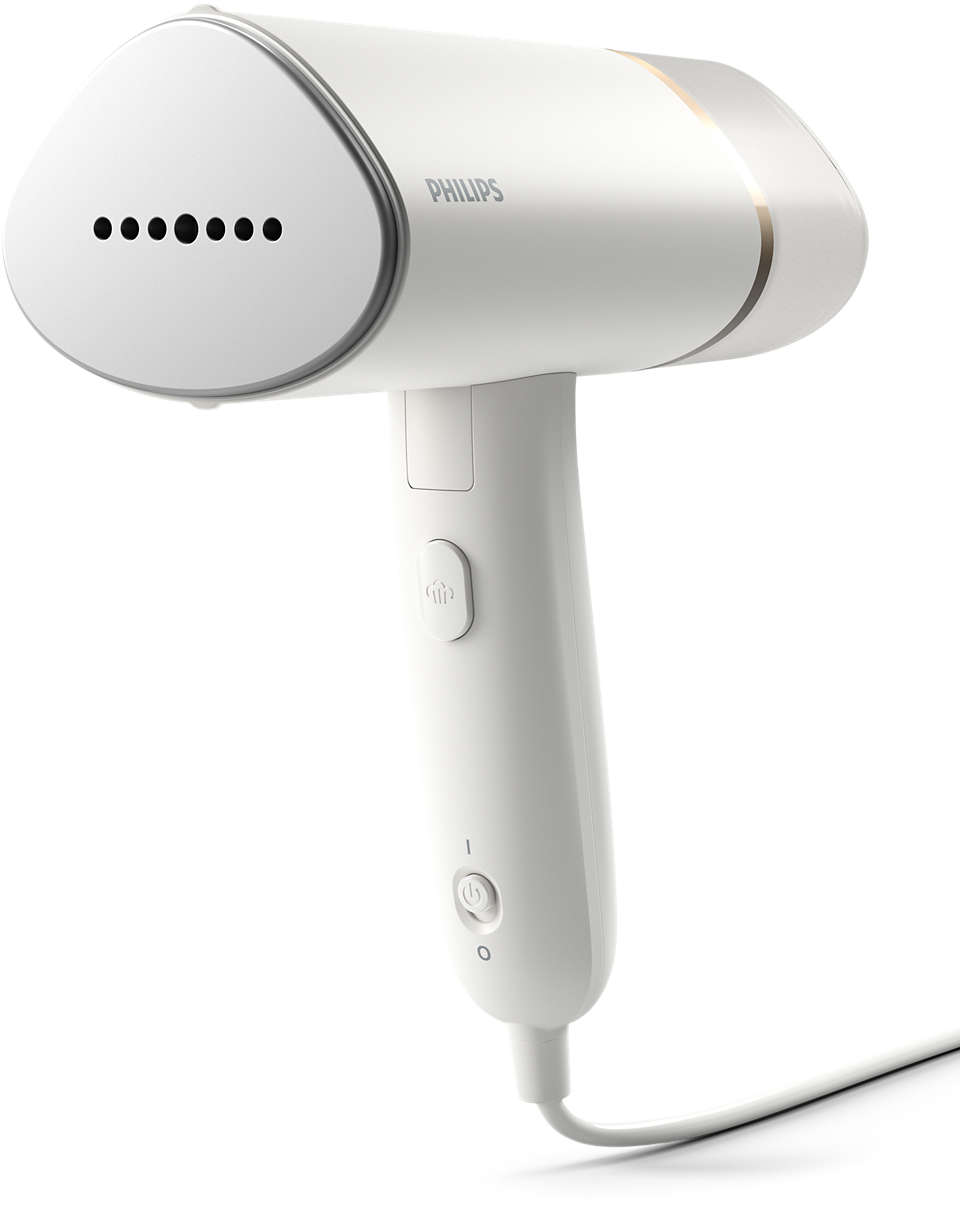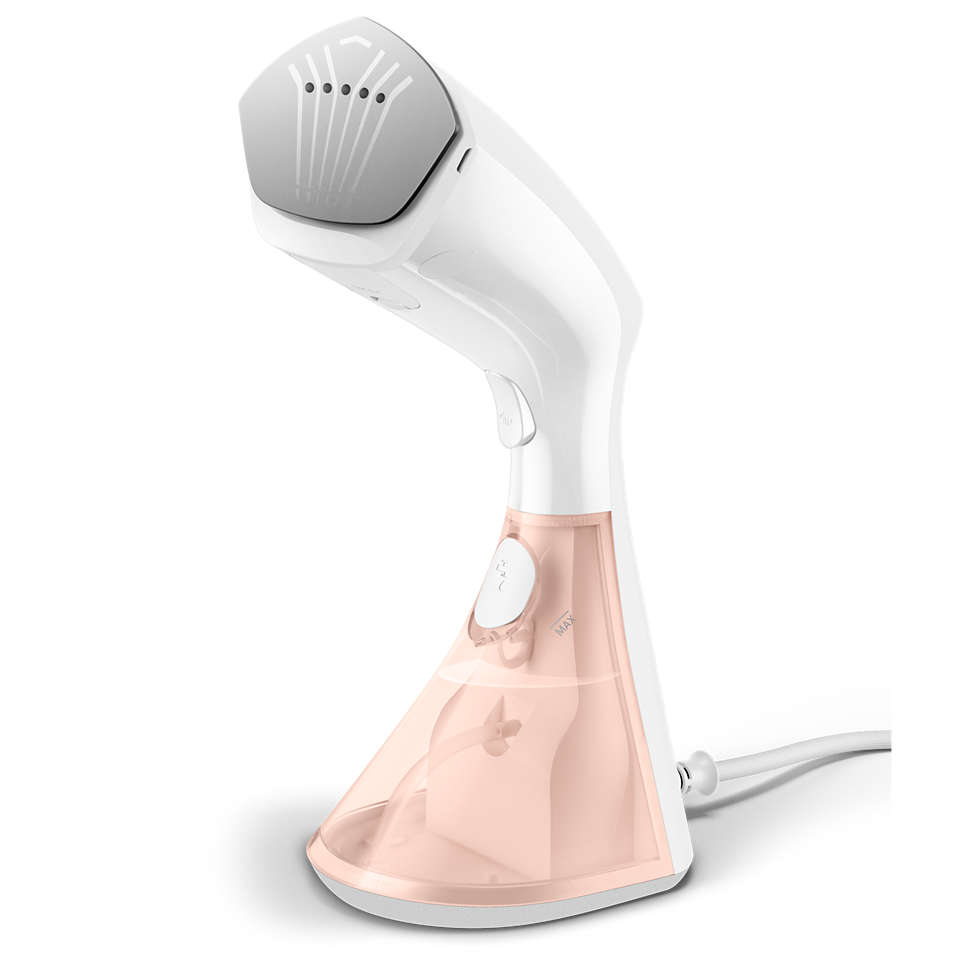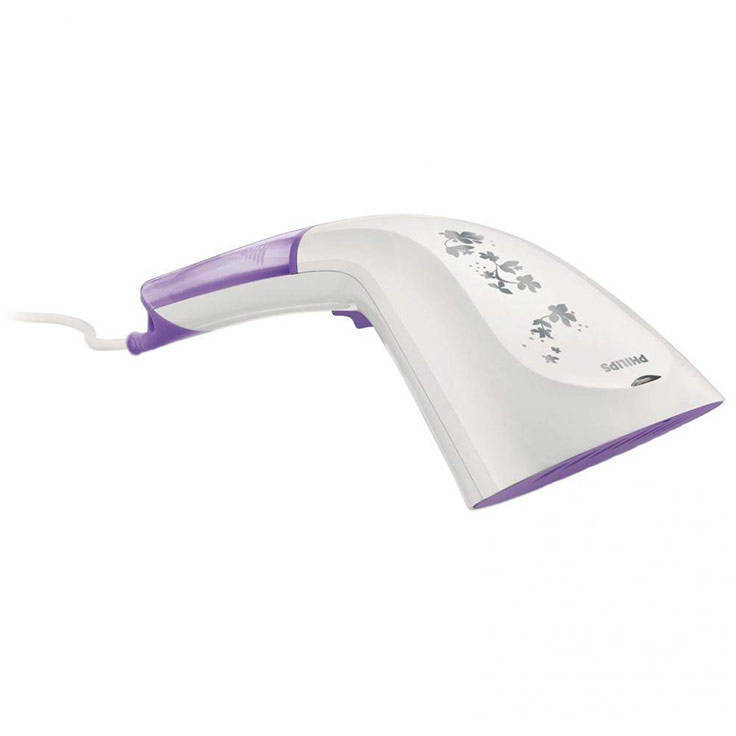
Philips Aparat de calcat vertical cu abur Steam & Go GC320/55, 1200 W, 0.06 l, 24 g/min, alb/mov - Pret: 0,00 lei - Badabum.ro
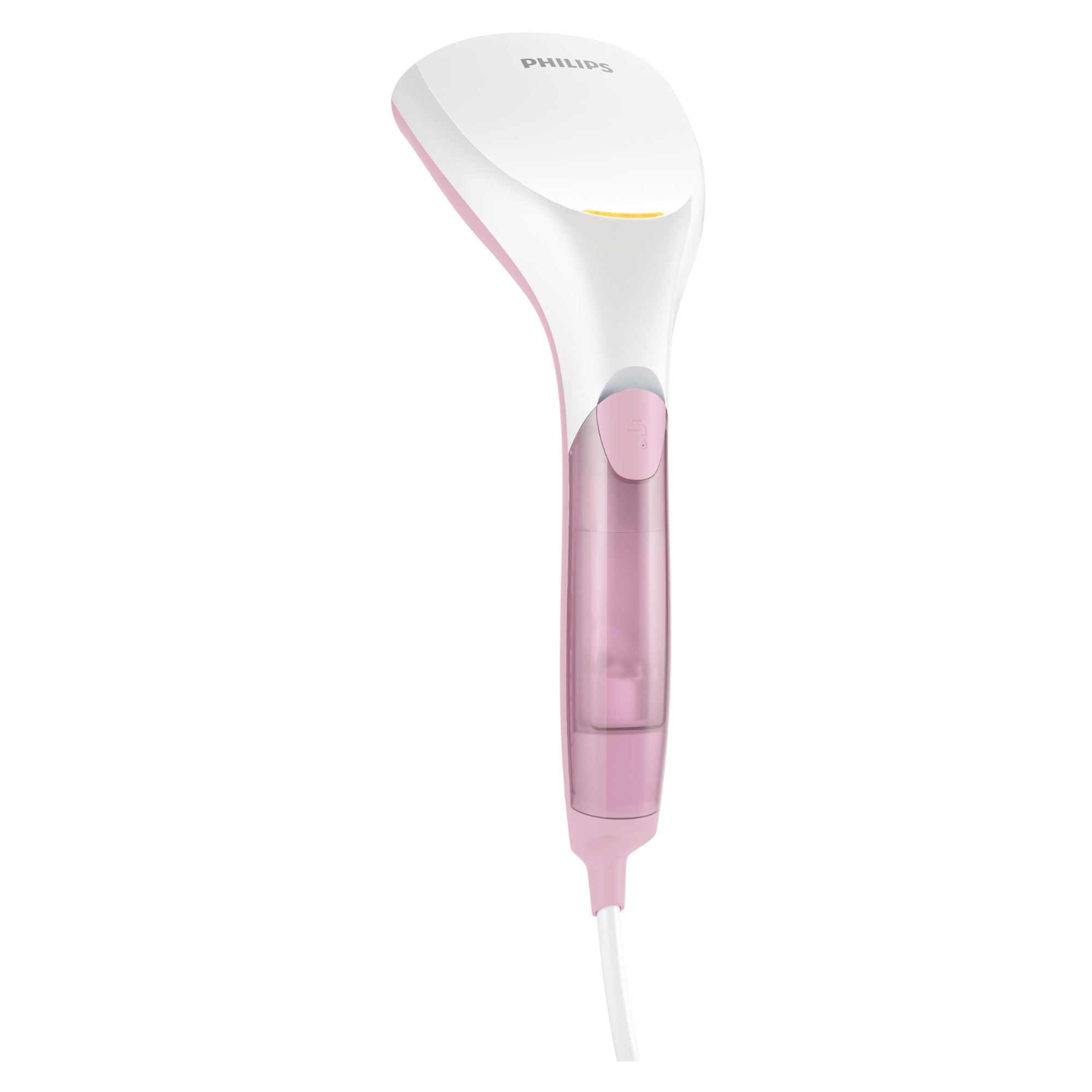
Dedeman - Aparat de calcat vertical cu abur Philips GC299/40, 1000 W, 0.06 l, 20 g/min, jet de abur continuu, perie pentru materiale groase, alb cu roz - Dedicat planurilor tale